Scalable Industrial Preparation of High-Purity Palbociclib Intermediates via Novel Catalytic Hydrogenation
Introduction to Advanced Intermediate Manufacturing
The escalating global demand for Palbociclib, a potent CDK4/6 inhibitor used in advanced breast cancer therapy, has placed immense pressure on the supply chains of its key precursors. Central to this supply chain is Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate, a critical building block whose quality directly impacts the safety and efficacy of the final drug substance. Recent intellectual property developments, specifically patent CN113636973B, have unveiled a transformative industrial preparation method that addresses long-standing challenges regarding purity, color, and cost-efficiency. This novel methodology shifts away from expensive protected starting materials, opting instead for a robust acid-catalyzed nucleophilic substitution followed by precise catalytic hydrogenation. For procurement leaders and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding the technical nuances of this patent is essential for securing a competitive advantage in API manufacturing.
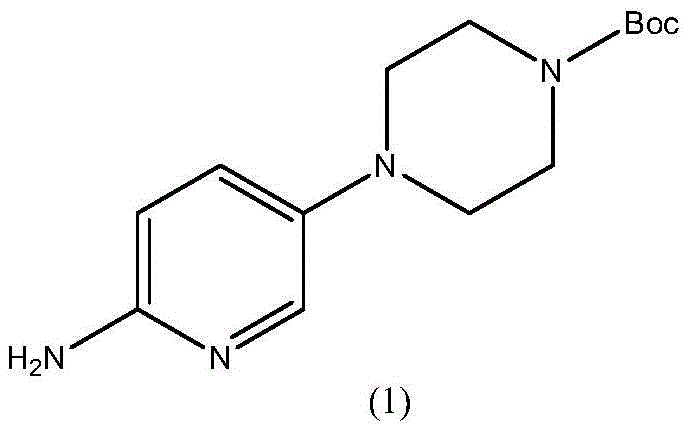
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this pivotal intermediate has been plagued by economic and technical inefficiencies inherent in traditional routes. One prominent literature method involves the coupling of 5-bromo-2-nitropyridine with 1-Boc-piperazine. While chemically straightforward, this approach suffers from severe cost penalties because 1-Boc-piperazine itself is an expensive reagent with low atom economy; its preparation generates significant waste and often contains di-Boc impurities that complicate downstream purification. Furthermore, alternative routes utilizing 5-chloro-2-nitropyridine and free piperazine under alkaline conditions frequently encounter issues with over-alkylation. In these basic environments, the high nucleophilicity of the piperazine nitrogen leads to the formation of bimolecular condensation by-products, specifically piperazine disubstituted impurities, which are notoriously difficult to separate from the desired mono-substituted product.
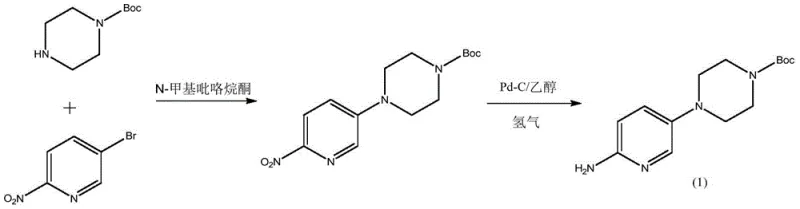
The Novel Approach
The breakthrough detailed in patent CN113636973B introduces a paradigm shift by utilizing 5-bromo-2-nitropyridine and unprotected piperazine in a mixed solvent system of n-butanol and water, catalyzed by strong acids like hydrobromic acid. This acidic environment is crucial as it protonates one nitrogen of the piperazine ring, effectively masking its nucleophilicity and ensuring highly selective mono-substitution at the other nitrogen. This strategic manipulation of reaction conditions virtually eliminates the formation of dimeric impurities at the source. Following the substitution, the process employs a biphasic Boc-protection strategy and a refined catalytic hydrogenation step using platinum carbon with an acid scavenger. This integrated approach not only simplifies the operational workflow but also ensures the production of a high-purity, light-colored solid suitable for stringent pharmaceutical applications, representing a significant leap forward for cost reduction in API manufacturing.
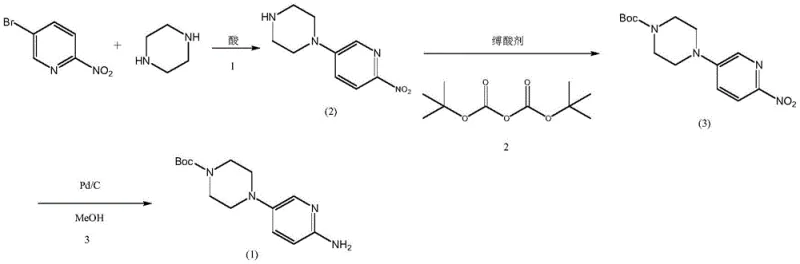
Mechanistic Insights into Acid-Catalyzed Selective Substitution
The core innovation of this technology lies in the precise control of piperazine reactivity through acid-base chemistry. In conventional alkaline syntheses, both nitrogen atoms of the piperazine ring are available for nucleophilic attack, leading to a statistical distribution of mono- and di-substituted products. However, by introducing a stoichiometric amount of hydrobromic acid, the process converts piperazine into its mono-hydrobromide salt. In this protonated state, one nitrogen atom carries a positive charge and is electronically deactivated towards nucleophilic aromatic substitution. This leaves only the unprotonated nitrogen available to attack the electron-deficient 5-bromo-2-nitropyridine. This mechanistic safeguard ensures that the reaction proceeds with exceptional regioselectivity, yielding the 1-(6-nitropyridin-3-yl)piperazine acid salt with minimal formation of the problematic bis-pyridyl piperazine impurities shown in Formula 4 and 5.
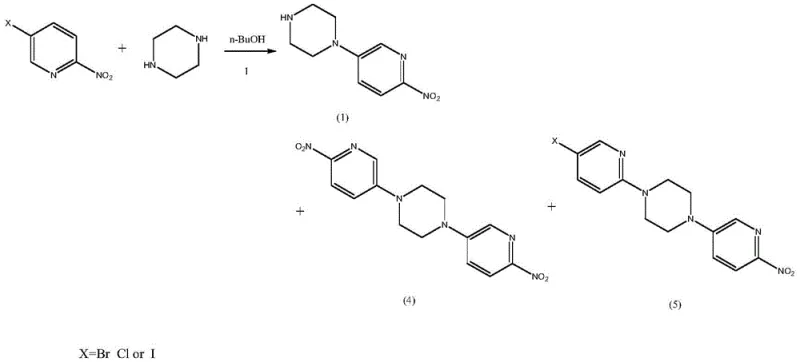
Furthermore, the patent addresses the critical stability of the Boc protecting group during the final reduction step. Catalytic hydrogenation of nitro groups often generates trace amounts of hydrobromic acid if debromination of residual halogenated impurities occurs. This localized acidity can catalyze the hydrolysis of the tert-butoxycarbonyl (Boc) group, leading to deprotected amines and potentially genotoxic urethane derivatives. To counteract this, the novel process incorporates a weak base, such as sodium acetate, directly into the hydrogenation mixture. This acid-binding agent acts as a buffer, neutralizing any generated HBr in situ before it can attack the carbamate linkage. This subtle yet powerful addition preserves the integrity of the Boc group, ensuring that the final product maintains its structural fidelity and meets rigorous safety standards without requiring complex downstream remediation.
How to Synthesize Tert-Butyl 4-(6-Aminopyridin-3-yl)Piperazine-1-Carboxylate Efficiently
The implementation of this industrial preparation method requires strict adherence to the optimized reaction parameters defined in the patent to maximize yield and purity. The process is designed to be telescoped where possible, minimizing isolation steps and solvent usage. The initial substitution is conducted at moderate temperatures to balance reaction rate with selectivity, followed by a unique solvent recovery system via azeotropic distillation that allows for the recycling of n-butanol. The subsequent protection and reduction steps utilize common, scalable reagents but rely on specific molar ratios and pH controls to prevent side reactions. For R&D teams looking to replicate or license this technology, the following standardized synthesis steps outline the critical operational sequence required to achieve the reported commercial-grade quality.
- React 5-bromo-2-nitropyridine with piperazine in n-butanol/water using hydrobromic acid catalyst to form the acid salt, then isolate the free base.
- Protect the piperazine nitrogen by reacting the wet intermediate with Boc anhydride in a toluene/water biphasic system using sodium carbonate.
- Perform catalytic hydrogenation of the nitro group using platinum carbon in methanol with sodium acetate as an acid scavenger.
- Purify the crude amine by dissolving in weak acid, decolorizing with activated carbon, and recrystallizing from alkaline solution.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that extend beyond simple chemical yield, addressing critical pain points in modern pharmaceutical supply chains. By eliminating the need for pre-purchased 1-Boc-piperazine, manufacturers can drastically reduce raw material costs, as unprotected piperazine is a commodity chemical available at a fraction of the price of its protected counterpart. Additionally, the ability to recycle the n-butanol solvent through azeotropic distillation significantly lowers both material consumption and waste disposal costs, contributing to a more sustainable and economically viable production model. The simplified purification protocol, which avoids complex chromatographic separations, further enhances throughput and reduces the overall manufacturing cycle time, ensuring a more responsive supply capability for downstream API producers.
- Cost Reduction in Manufacturing: The substitution of expensive protected starting materials with commodity piperazine and the implementation of solvent recycling loops create a substantially leaner cost structure. By avoiding the purchase of high-cost 1-Boc-piperazine and minimizing the loss of organic solvents through efficient recovery systems, the overall cost of goods sold is significantly optimized. Furthermore, the high selectivity of the acid-catalyzed reaction reduces the burden on purification resources, lowering the consumption of adsorbents and recrystallization solvents typically required to remove stubborn impurities.
- Enhanced Supply Chain Reliability: Relying on widely available bulk chemicals like 5-bromo-2-nitropyridine and piperazine mitigates the risk of supply disruptions often associated with specialized, low-volume reagents. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without compromising product quality, ensures consistent batch-to-batch performance. This reliability is crucial for maintaining continuous API production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, utilizing standard unit operations such as filtration, distillation, and crystallization that are easily transferable from pilot to commercial plants. The reduction in hazardous waste generation, particularly through the minimization of Boc-anhydride byproducts and the recycling of aqueous phases, aligns with increasingly stringent environmental regulations. This green chemistry approach not only reduces the environmental footprint but also simplifies regulatory compliance and waste management logistics for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these details is vital for technical teams evaluating the feasibility of adopting this process for large-scale production.
Q: How does the new process prevent the formation of piperazine disubstituted impurities?
A: The process utilizes an acid-catalyzed mono-salt formation strategy where piperazine is partially protonated. This protects one amino group, effectively blocking bimolecular nucleophilic attack and preventing the formation of difficult-to-remove dimeric by-products common in alkaline conditions.
Q: Why is an acid-binding agent added during the hydrogenation step?
A: Trace debromination of impurities during hydrogenation can generate hydrobromic acid, which catalyzes the hydrolysis of the sensitive Boc protecting group. Adding sodium acetate neutralizes this acid in situ, preserving the Boc group and preventing the formation of genotoxic impurities.
Q: What are the purity and color specifications achievable with this method?
A: By employing a specific activated carbon decolorization step in weak acid followed by alkaline crystallization, the method consistently yields a white to off-white solid with HPLC purity exceeding 99.8%, significantly improving upon the brown-yellow solids reported in prior art.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyl 4-(6-Aminopyridin-3-yl)Piperazine-1-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs like Palbociclib depends on the uninterrupted supply of high-quality intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners. We are committed to delivering products with stringent purity specifications, leveraging advanced analytical capabilities in our rigorous QC labs to guarantee that every batch meets the highest standards of safety and consistency required for clinical and commercial use.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized manufacturing processes can drive value for your organization. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey towards efficient and compliant API manufacturing.
