Advanced Synthesis of High-Purity Amide Derivatives for Neurological Drug Manufacturing
Advanced Synthesis of High-Purity Amide Derivatives for Neurological Drug Manufacturing
The pharmaceutical landscape for treating severe neurological disorders, particularly schizophrenia, demands intermediates of exceptional purity and structural integrity. Recent intellectual property developments, specifically patent CN113195454A, have introduced a groundbreaking preparation method for amide-like derivatives represented by Formula VI. This compound serves as a critical precursor for dopamine D2 and serotonin receptor modulators, which are essential in managing psychiatric conditions affecting millions globally. The disclosed technology addresses long-standing challenges in the synthesis of these complex heterocyclic systems, offering a pathway that significantly enhances product quality while mitigating the environmental and safety hazards associated with traditional manufacturing protocols. For R&D directors and procurement specialists, understanding the nuances of this new route is vital for securing a reliable supply chain of high-grade active pharmaceutical ingredients.
This technical insight report dissects the innovative synthetic strategy outlined in the patent, highlighting its superiority over existing methodologies. By leveraging advanced Lewis acid catalysis and optimized phase transfer conditions, the new process achieves a purity profile that meets stringent pharmaceutical standards, often exceeding 99%. This level of refinement is not merely a technical achievement but a commercial imperative, as it directly correlates to reduced purification costs and accelerated regulatory approval timelines for the final drug product. As we delve deeper into the mechanistic and operational details, the value proposition for integrating this technology into your supply chain becomes increasingly clear.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in patent WO2017084627A, have historically relied on aggressive chemical transformations that pose significant operational risks. A primary bottleneck in conventional synthesis is the demethylation step, which typically utilizes refluxing hydrobromic acid (HBr). This reagent is highly corrosive, generating toxic gases that necessitate expensive scrubbing systems and specialized glass-lined or Hastelloy reactors to prevent equipment failure. Furthermore, the alkylation steps in older routes often employ 1,3-dibromopropane, a reagent prone to forming bis-alkylated byproducts and other difficult-to-remove impurities. These side reactions compromise the overall yield and create a complex impurity spectrum that requires extensive and costly chromatographic purification to resolve.
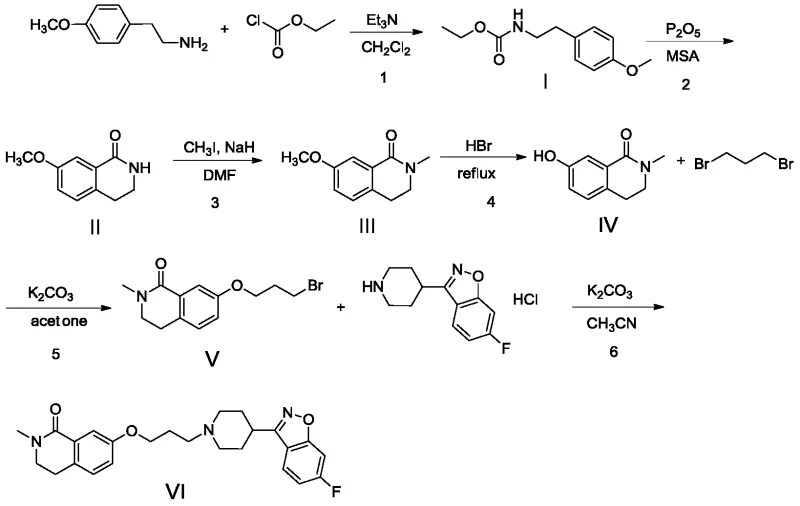
The cumulative effect of these inefficiencies is a manufacturing process that is not only environmentally burdensome but also economically unsustainable at scale. The high impurity load in the crude product, often resulting in purities around 92%, forces manufacturers to accept lower yields after rigorous recrystallization or column chromatography. For supply chain managers, this translates to unpredictable lead times and higher cost-of-goods-sold (COGS). The reliance on hazardous reagents also complicates waste disposal and increases the regulatory compliance burden, making the conventional route less attractive for modern, green chemistry-focused production facilities seeking to minimize their environmental footprint.
The Novel Approach
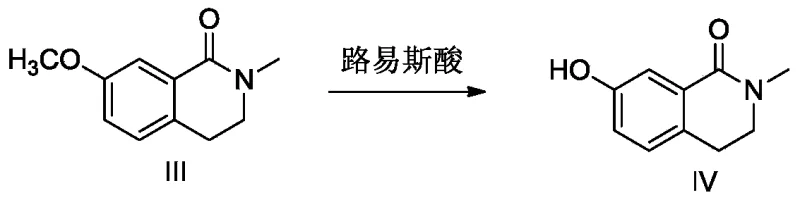
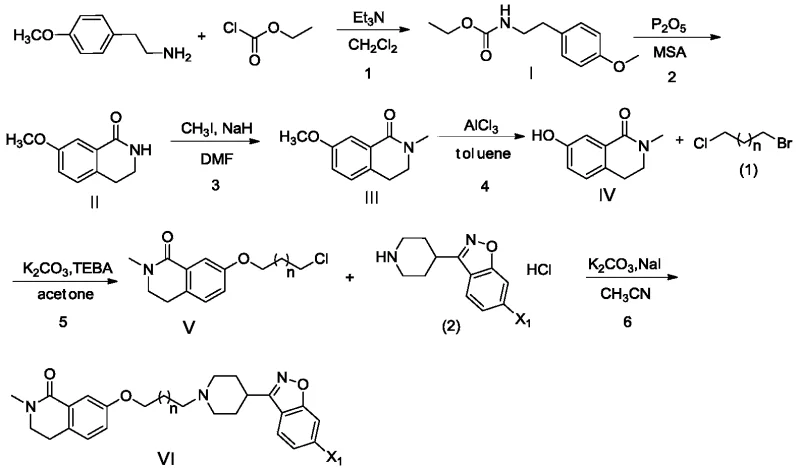
In stark contrast, the methodology presented in CN113195454A introduces a paradigm shift by replacing hazardous reagents with safer, more selective alternatives. The most notable innovation is the substitution of hydrobromic acid with aluminum chloride (AlCl3) or ferric trichloride for the demethylation of the methoxy intermediate. This Lewis acid-mediated cleavage occurs under controlled conditions in organic solvents like toluene, eliminating the evolution of corrosive HBr gas and significantly improving operator safety. Additionally, the process utilizes 1-bromo-3-chloropropane instead of 1,3-dibromopropane for the etherification step. The difference in leaving group ability between bromine and chlorine provides superior regioselectivity, effectively suppressing the formation of bis-alkylated impurities and ensuring a cleaner reaction profile.
The impact of these modifications on the final product quality is profound. By minimizing side reactions at the source, the new route delivers a crude product with significantly higher purity, which simplifies the downstream purification process. The patent data indicates that this approach can consistently achieve purities greater than 99%, a benchmark that is difficult to reach with legacy methods without excessive material loss. For procurement teams, this means a more robust supply of intermediates that require less rework, leading to substantial cost reductions in the overall manufacturing budget. The enhanced operability and reduced environmental pressure make this novel approach an ideal candidate for scaling up to meet the growing global demand for neurological therapeutics.
Mechanistic Insights into Lewis Acid-Catalyzed Demethylation and Etherification
The core of this technological advancement lies in the precise control of reaction mechanisms to favor the desired product while suppressing degradation pathways. The demethylation step utilizing aluminum chloride proceeds through a coordination mechanism where the Lewis acid binds to the oxygen atom of the methoxy group, weakening the carbon-oxygen bond and facilitating nucleophilic attack by chloride ions. Unlike the radical-heavy environment of HBr reflux, this ionic pathway is cleaner and generates fewer chlorinated byproducts on the aromatic ring. The choice of toluene as a solvent is critical, as it provides a high boiling point sufficient to drive the reaction to completion while maintaining the solubility of the intermediate complexes, ensuring homogeneous reaction conditions that are essential for batch consistency.
Furthermore, the subsequent etherification reaction leverages the principles of phase transfer catalysis (PTC) to enhance efficiency. By employing quaternary ammonium salts like benzyltriethylammonium chloride (TEBA), the reaction facilitates the transport of the phenoxide anion into the organic phase where it reacts with the alkyl halide. This catalytic cycle allows the use of mild inorganic bases like potassium carbonate instead of strong, moisture-sensitive hydrides, thereby reducing safety risks and cost. The specific selection of 1-bromo-3-chloropropane exploits the kinetic difference between the two halogens; the bromine acts as the primary leaving group for the initial ether bond formation, while the chlorine remains intact for the subsequent coupling with the piperidine fragment. This orthogonal reactivity is key to preventing polymerization and ensuring high selectivity.
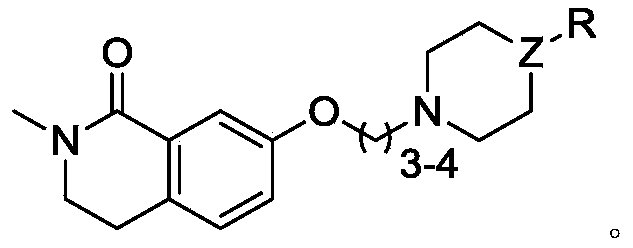
How to Synthesize Formula VI Intermediate Efficiently
The synthesis of the target amide derivative is a multi-step sequence that begins with the construction of the isoquinolinone core and concludes with the attachment of the pharmacophore side chain. The process starts with the protection of 4-methoxyphenethylamine as a carbamate, followed by cyclization using phosphorus pentoxide in methanesulfonic acid to form the lactam ring. Subsequent N-methylation yields the methoxy-intermediate, which then undergoes the critical Lewis acid demethylation described earlier. The resulting phenol is alkylated to introduce the propyl linker, and finally, the piperidine-isoxazole moiety is attached via nucleophilic substitution. Each step has been optimized for yield and purity, creating a cohesive workflow that minimizes isolation losses.
- Synthesize intermediate III via carbamate formation, cyclization with P2O5/MSA, and N-methylation using CH3I/NaH.
- Perform Lewis acid demethylation on intermediate III using AlCl3 in toluene to obtain phenolic intermediate IV.
- React intermediate IV with 1-bromo-3-chloropropane using TEBA catalyst, followed by coupling with the piperidine-isoxazole fragment to yield Formula VI.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the adoption of this novel synthesis route offers compelling economic and logistical benefits. The shift away from corrosive and toxic reagents like hydrobromic acid directly translates to lower capital expenditure on specialized corrosion-resistant equipment and reduced maintenance downtime. Furthermore, the elimination of hazardous gas scrubbing requirements simplifies facility compliance and lowers the total cost of ownership for the production line. The improved selectivity of the reagents means that less raw material is wasted on byproduct formation, leading to a more efficient utilization of inputs and a reduction in the volume of chemical waste that requires treatment and disposal.
- Cost Reduction in Manufacturing: The streamlined purification process resulting from higher crude purity significantly lowers the consumption of solvents and adsorbents typically required for chromatography. By avoiding the formation of difficult-to-remove impurities, the new method reduces the number of recrystallization cycles needed, which in turn decreases energy consumption and labor hours. The use of inexpensive inorganic bases and common organic solvents further drives down the variable costs per kilogram, making the final intermediate more price-competitive in the global market without sacrificing quality standards.
- Enhanced Supply Chain Reliability: The robustness of the new chemical process ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted drug production schedules. The reliance on commercially available and stable reagents like aluminum chloride and 1-bromo-3-chloropropane mitigates the risk of supply disruptions often associated with specialized or hazardous chemicals. This stability allows for better inventory planning and reduces the need for excessive safety stock, thereby optimizing working capital and ensuring that downstream API manufacturing can proceed without unexpected delays caused by intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily managed in large-scale reactors without exothermic runaways or pressure spikes. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, future-proofing the supply chain against potential regulatory crackdowns on toxic effluents. This green chemistry advantage not only protects the company's reputation but also facilitates smoother regulatory audits and approvals, accelerating the time-to-market for new pharmaceutical products derived from these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: How does the new synthesis method improve product purity compared to prior art?
A: The novel method replaces hazardous HBr demethylation with AlCl3 and utilizes 1-bromo-3-chloropropane instead of 1,3-dibromopropane. This strategic change significantly reduces side reactions and unknown impurities, achieving HPLC purity exceeding 99.7%, whereas conventional methods often struggle to reach 93%.
Q: What are the safety advantages of using Aluminum Chloride over Hydrobromic Acid?
A: Traditional demethylation using refluxing hydrobromic acid generates toxic HBr gas and requires specialized corrosion-resistant equipment. The patented Lewis acid approach using AlCl3 in toluene operates under milder conditions, drastically reducing operator exposure risks and lowering maintenance costs for reactor vessels.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It avoids extremely harsh conditions, utilizes common solvents like acetone and toluene, and employs robust phase transfer catalysts like TEBA, ensuring consistent yields and simplified downstream processing suitable for metric ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formula VI Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving neurological medications. Our team of expert chemists has thoroughly analyzed the synthetic pathway described in CN113195454A and possesses the technical capability to execute this advanced route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Formula VI intermediate we deliver meets the highest pharmaceutical grade standards required for human use.
We invite you to collaborate with us to leverage this innovative technology for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall production costs.
