Advanced Manufacturing of Relugolix Intermediates Using Non-Precious Metal Catalysis for Global Pharma Supply
Introduction to Next-Generation Relugolix Manufacturing
The pharmaceutical landscape for gonadotropin-releasing hormone (GnRH) antagonists has been significantly advanced by the development of Relugolix, a potent oral small molecule indicated for treating uterine leiomyoma. However, the commercial viability of such complex APIs often hinges on the efficiency and safety of their synthetic routes. Patent CN113501830A introduces a groundbreaking preparation method that fundamentally reengineers the synthesis of Relugolix and its critical intermediates, specifically targeting the reduction of nitro groups and the formation of urea linkages. This innovation addresses long-standing industry pain points regarding heavy metal contamination and process safety.
Traditionally, the synthesis of the core thiophene-pyrimidine scaffold relied heavily on precious metal catalysts. The new methodology described in this patent replaces these costly and hazardous reagents with an inexpensive iron-based catalytic system. By utilizing ferric chloride and activated carbon in conjunction with hydrazine hydrate, the process achieves high conversion rates while completely bypassing the need for high-pressure hydrogenation. This shift not only enhances operational safety but also simplifies the downstream purification workflow, ensuring that the final active pharmaceutical ingredient meets stringent global regulatory standards for residual metals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in Chinese patent applications CN200480008798.9 and CN201380051107.2, have historically depended on palladium-carbon or platinum-carbon catalysts for the critical nitro reduction steps. These conventional approaches present substantial logistical and financial burdens for large-scale manufacturers. The primary drawback is the inherent risk of heavy metal residues; removing trace amounts of palladium or platinum to meet pharmacopeial limits often requires complex scavenging steps or repeated recrystallizations, which drastically reduce overall yield. Furthermore, the reliance on hydrogen gas necessitates specialized high-pressure reactors, introducing significant safety hazards related to pressurization and potential leaks in a production environment.
Beyond safety and cost, the chemical efficiency of these older routes is often compromised by the formation of difficult-to-remove impurities. For instance, the condensation of arylamines with O-methylhydroxylamine in the presence of carbonyldiimidazole (CDI) in previous methods frequently leads to the generation of dimeric impurities. These byproducts possess similar physicochemical properties to the target molecule, making their separation via standard chromatography both tedious and expensive. The cumulative effect of these inefficiencies is a process that is ill-suited for the robust, cost-sensitive demands of modern generic and branded pharmaceutical supply chains.
The Novel Approach
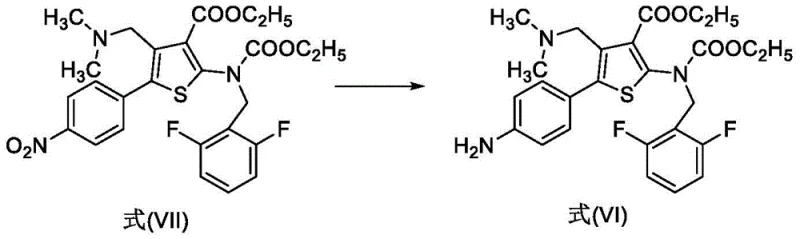
The innovative route presented in patent CN113501830A offers a transformative solution by substituting noble metals with a ferric chloride and activated carbon catalytic system. This non-precious metal approach effectively mediates the reduction of the nitro group in Formula VII to the corresponding aniline in Formula VI under mild thermal conditions. The elimination of palladium not only removes the risk of toxic metal residues but also decouples the process from the volatile pricing of precious metals, providing a more stable cost structure for procurement teams. Additionally, the reaction proceeds at atmospheric pressure, allowing facilities to utilize standard glass-lined or stainless steel reactors without the need for expensive hydrogenation infrastructure.
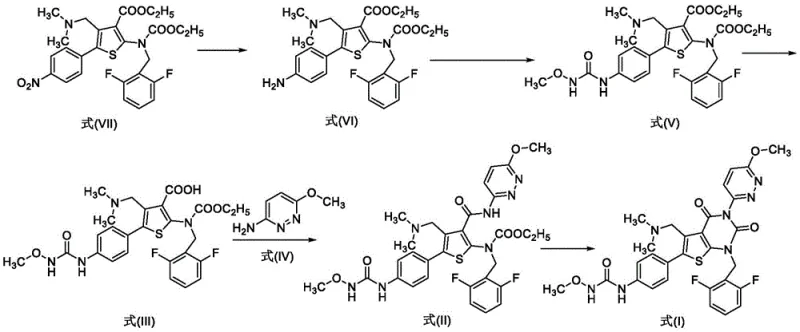
Moreover, the novel process incorporates strategic crystallization steps that serve as powerful purification tools. Specifically, during the conversion of Formula VI to Formula V, the protocol employs ethyl acetate as a crystallization solvent. This specific solvent choice is engineered to selectively precipitate the desired urea intermediate while keeping dimeric impurities in the mother liquor. This capability to control the impurity profile through physical separation rather than chemical scavenging represents a significant leap forward in process chemistry, enabling the production of high-purity intermediates suitable for direct progression to the final cyclization step without intermediate chromatographic purification.
Mechanistic Insights into Iron-Catalyzed Nitro Reduction
The core mechanistic advantage of this new synthesis lies in the electron transfer mediation facilitated by the iron species. In the reduction of Formula VII to Formula VI, ferric chloride acts as a Lewis acid and electron shuttle in the presence of hydrazine hydrate. The activated carbon serves as a support that enhances the surface area for the reaction, promoting efficient contact between the nitro substrate and the reducing agent. Unlike heterogeneous hydrogenation where hydrogen gas must diffuse into the catalyst pores, this liquid-phase reduction allows for homogeneous-like kinetics while maintaining the ease of filtration associated with heterogeneous systems. The reaction temperature is carefully controlled between 50°C and 80°C, a range that optimizes the reaction rate while minimizing the thermal degradation of the sensitive thiophene ester moieties.
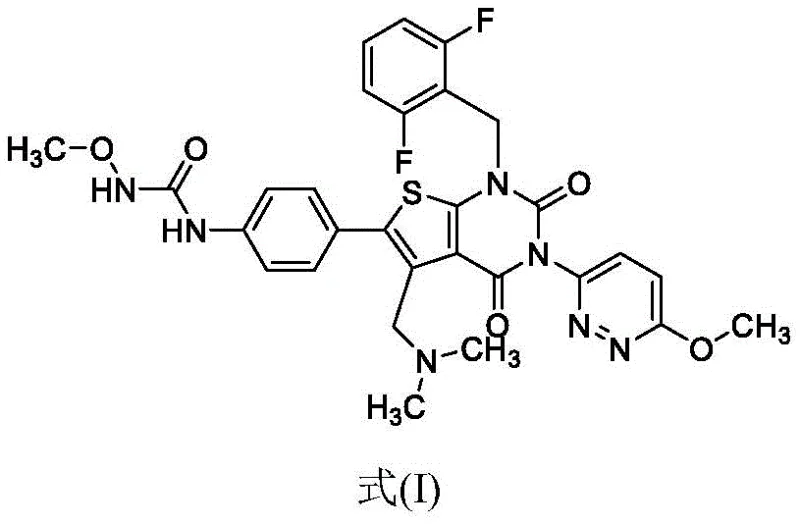
Impurity control is further refined in the subsequent amidation and cyclization stages. The conversion of the carboxylic acid intermediate (Formula III) to the amide (Formula II) and finally to the cyclic Relugolix (Formula I) is managed through precise stoichiometric control of coupling reagents like propylphosphonic anhydride. The mechanism ensures that the nucleophilic attack of the pyridazine amine occurs selectively at the activated carboxyl group, preventing side reactions at the urea nitrogen. The final intramolecular cyclization to form the thieno[2,3-d]pyrimidine ring is driven by the basicity of sodium methoxide, which deprotonates the urea nitrogen, facilitating the nucleophilic displacement of the ethoxycarbonyl leaving group. This cascade of reactions is designed to proceed with minimal byproduct formation, ensuring that the final API achieves a purity profile exceeding 99.8%.
How to Synthesize Relugolix Intermediates Efficiently
The synthesis of Relugolix intermediates via this novel route involves a streamlined five-step sequence that prioritizes operational simplicity and yield maximization. The process begins with the reduction of the nitro precursor, followed by urea formation, ester hydrolysis, amidation, and final cyclization. Each step has been optimized to utilize common industrial solvents such as ethanol, acetonitrile, and ethyl acetate, avoiding exotic or highly toxic reagents. The following guide outlines the critical operational parameters derived from the patent examples, focusing on the transformation of the nitro-thiophene scaffold into the advanced amine intermediate.
- React compound Formula VII with hydrazine hydrate, ferric chloride, and activated carbon in ethanol at 50-80°C to obtain Formula VI.
- React Formula VI with methoxy ammonium salt, CDI, and base in acetonitrile, followed by crystallization in ethyl acetate to yield Formula V.
- Hydrolyze Formula V with potassium hydroxide in ethanol, followed by crystallization to obtain the carboxylic acid intermediate Formula III.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed synthesis route offers compelling economic and logistical benefits. The most immediate impact is the drastic reduction in raw material costs associated with catalyst consumption. By eliminating the need for palladium on carbon, which is subject to significant market price fluctuations and supply constraints, manufacturers can stabilize their bill of materials. Furthermore, the removal of high-pressure hydrogenation steps reduces the capital expenditure required for specialized reactor vessels and safety systems, allowing existing multipurpose facilities to produce these high-value intermediates without major retrofitting investments.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with inexpensive iron salts results in a substantial decrease in direct material costs. Since the process does not require expensive metal scavengers or complex purification columns to remove heavy metals, the operational expenditure per kilogram of product is significantly lowered. This cost efficiency is compounded by the high yields reported in the patent examples, which minimize waste and maximize the throughput of valuable starting materials.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like ferric chloride and hydrazine hydrate ensures a robust and resilient supply chain. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents used in this process are widely available from multiple global suppliers. This diversification mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on atmospheric pressure and standard thermal conditions. The absence of high-pressure hydrogen gas eliminates a major safety hazard, simplifying regulatory compliance and insurance requirements for manufacturing sites. Additionally, the use of recyclable solvents and the generation of less hazardous waste streams align with green chemistry principles, supporting corporate sustainability goals and reducing the environmental footprint of pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Relugolix synthesis route. These insights are derived directly from the experimental data and beneficial effects described in patent CN113501830A, providing clarity on how this technology compares to established industry standards.
Q: Why is the iron-catalyzed reduction method superior to traditional palladium-carbon hydrogenation for Relugolix synthesis?
A: The iron-catalyzed method eliminates the need for expensive noble metals like palladium or platinum, significantly reducing raw material costs. Furthermore, it avoids the safety hazards associated with high-pressure hydrogenation equipment and prevents the risk of heavy metal residues exceeding regulatory limits in the final API.
Q: How does the new process control dimeric impurities during the urea formation step?
A: The process utilizes specific crystallization conditions involving ethyl acetate as a solvent after the reaction of Formula VI with methoxyamine salts and CDI. This targeted purification strategy efficiently removes dimeric impurities that are commonly generated in prior art methods, ensuring high purity without complex chromatography.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability. It operates under atmospheric pressure without specialized high-pressure reactors, uses common solvents like ethanol and acetonitrile, and relies on simple filtration and crystallization for purification, making it highly adaptable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Relugolix Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the iron-catalyzed route for Relugolix intermediates and is fully prepared to implement this methodology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective and safe synthesis route can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to validate the superior quality and economic advantages of our Relugolix intermediates, ensuring a reliable partnership for your long-term drug development needs.
