Advanced One-Step Synthesis of Exocyclic Double Bond Quinoline Derivatives for Commercial Scale-Up
Introduction to Novel Quinoline Scaffold Construction
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the demand for more efficient routes to complex pharmacophores. Patent CN110172076B introduces a groundbreaking methodology for the construction of quinoline derivatives containing exocyclic double bonds, a structural motif frequently encountered in bioactive molecules and advanced materials. This technology leverages a sophisticated cascade reaction sequence initiated by a Hexadehydro-Diels-Alder (HDDA) transformation, enabling the rapid assembly of highly functionalized quinoline cores from relatively simple tetrayne precursors. Unlike traditional methods that often require multiple discrete steps and harsh conditions, this approach integrates cyclization and functionalization into a streamlined thermal process. The utilization of a chiral ferrocene auxiliary not only facilitates the reaction but also imparts potential stereochemical value to the final products, making this route particularly attractive for the development of chiral pharmaceutical intermediates. 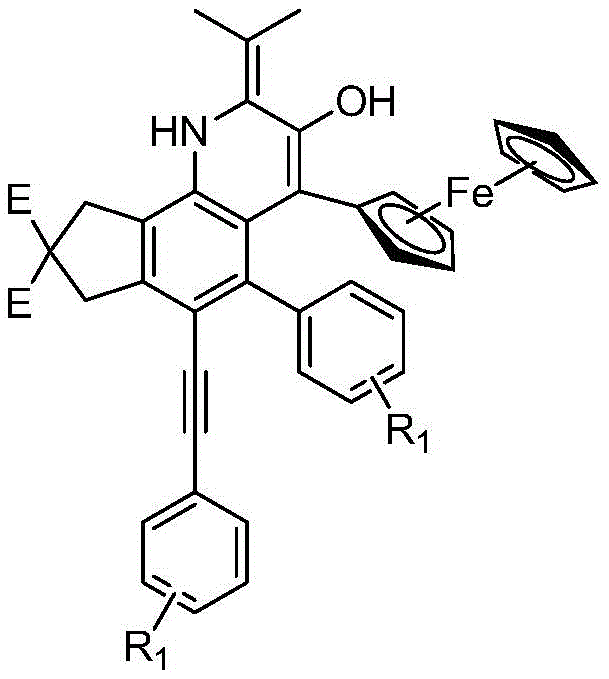
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline derivatives has relied heavily on classical condensations such as the Skraup, Doebner-Miller, or Friedlander reactions, which, while robust, often suffer from significant limitations regarding substrate scope and functional group tolerance. These traditional pathways frequently necessitate the use of strong acids or oxidants, leading to poor atom economy and the generation of substantial hazardous waste streams that complicate downstream processing. Furthermore, constructing quinolines with specific exocyclic unsaturation patterns typically requires additional post-functionalization steps, such as Wittig olefinations or elimination reactions, which drastically increase the overall step count and reduce the cumulative yield. The inability of these legacy methods to easily incorporate complex chiral fragments or sensitive organometallic motifs, such as ferrocene units, restricts the chemical diversity accessible to medicinal chemists exploring new biological targets. Consequently, the industry has long sought a more direct and versatile strategy that can bypass these inefficiencies while maintaining high purity standards.
The Novel Approach
The methodology disclosed in the patent data represents a paradigm shift by employing a tetrayne compound as a key synthon, which undergoes an intramolecular cyclization to generate a transient benzyne species under thermal conditions. This reactive intermediate is immediately trapped by the nitrogen lone pair of a chiral ferrocene-oxazoline derivative, initiating a cascade that constructs the quinoline ring system in a single operational step. This strategy eliminates the need for pre-functionalized aniline or aldehyde starting materials, thereby simplifying the supply chain requirements for raw materials. The reaction proceeds in toluene at moderate temperatures ranging from 100°C to 110°C, conditions that are easily manageable in standard glass-lined reactors without the need for specialized high-pressure equipment. By merging the ring formation and the installation of the exocyclic double bond into one pot, this novel approach significantly enhances the overall atom economy and reduces the solvent consumption associated with intermediate isolations. 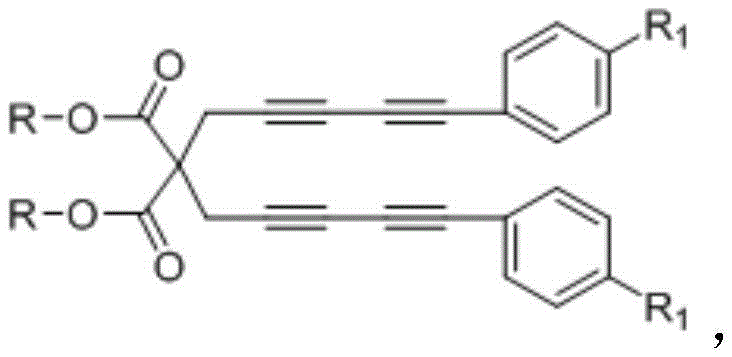
Mechanistic Insights into HDDA-Triggered Cascade Cyclization
The core of this synthetic innovation lies in the intricate mechanistic pathway that transforms the linear tetrayne precursor into the rigid quinoline architecture. The process begins with a thermal Hexadehydro-Diels-Alder (HDDA) reaction, where the conjugated alkyne system within the precursor undergoes a [4+2] cycloaddition to form a highly strained benzyne intermediate. This transient species acts as a powerful electrophile, susceptible to nucleophilic attack by the nitrogen atom of the (S)-(4-isopropyloxazolin-2-yl)ferrocene reagent. Following this initial addition, the system undergoes a formal [2+2] cycloaddition to generate a four-membered azetidine-like ring, which is inherently unstable due to ring strain. The subsequent ring-opening event leads to a seven-membered zwitterionic intermediate featuring a negative charge on the nitrogen and a positive charge on the carbon adjacent to the ferrocene moiety. This charge separation drives a series of rapid rearrangements, including hydride shifts and bond migrations, ultimately collapsing the structure into the thermodynamically stable quinoline framework with the characteristic exocyclic double bond.
From an impurity control perspective, the specificity of the HDDA reaction provides a significant advantage, as the formation of the benzyne intermediate is highly dependent on the precise geometry of the tetrayne substrate, minimizing off-pathway polymerization. The use of the ferrocene auxiliary further directs the regioselectivity of the nucleophilic attack, ensuring that the substitution pattern on the quinoline ring is consistent and predictable. Any unreacted precursor or side products generated during the thermal treatment can be effectively removed through standard silica gel chromatography, utilizing a gradient of ethyl acetate in petroleum ether. The final crystallization step, typically performed using petroleum ether as the anti-solvent, serves as a critical purification barrier to remove trace metal residues and isomeric impurities, ensuring the final API intermediate meets stringent quality specifications. This mechanistic clarity allows process chemists to fine-tune reaction parameters, such as concentration and temperature, to maximize the yield of the desired isomer while suppressing potential decomposition pathways.
How to Synthesize Exocyclic Double Bond Quinolines Efficiently
The practical execution of this synthesis involves a two-stage protocol, beginning with the preparation of the tetrayne precursor followed by the key cyclization event. The precursor is synthesized via a sequential alkylation of dialkyl malonate with propargyl bromide, followed by a palladium-catalyzed Sonogashira coupling with aryl ethynyl bromides to install the necessary alkyne arms. Once the precursor is secured, it is subjected to thermal conditions in toluene with the chiral ferrocene reagent to effect the transformation. The detailed standardized operating procedures, including specific molar ratios, solvent volumes, and workup protocols required to replicate the high yields reported in the patent examples, are outlined in the technical guide below.
- Synthesize the tetrayne precursor by alkylating dialkyl malonate with propargyl bromide, followed by Sonogashira coupling with aryl ethynyl bromides.
- React the tetrayne precursor with (S)-(4-isopropyloxazolin-2-yl)ferrocene in toluene solvent at elevated temperatures (100-110°C) for 8-14 hours.
- Purify the resulting quinoline derivative through aqueous workup, column chromatography using ethyl acetate and petroleum ether, and final crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits related to cost efficiency and operational reliability. The consolidation of multiple bond-forming events into a single thermal step drastically reduces the number of unit operations required, which directly correlates to lower labor costs and reduced equipment occupancy time. By eliminating the need for cryogenic reagents or exotic catalysts that are prone to supply shortages, the manufacturing process becomes more resilient to market fluctuations and geopolitical disruptions. The reliance on commodity chemicals such as toluene, malonates, and simple alkyl halides ensures a stable and cost-effective raw material base that can be sourced from multiple global suppliers. Furthermore, the high atom economy of the cascade reaction minimizes the generation of waste byproducts, leading to significant savings in waste disposal fees and environmental compliance costs. 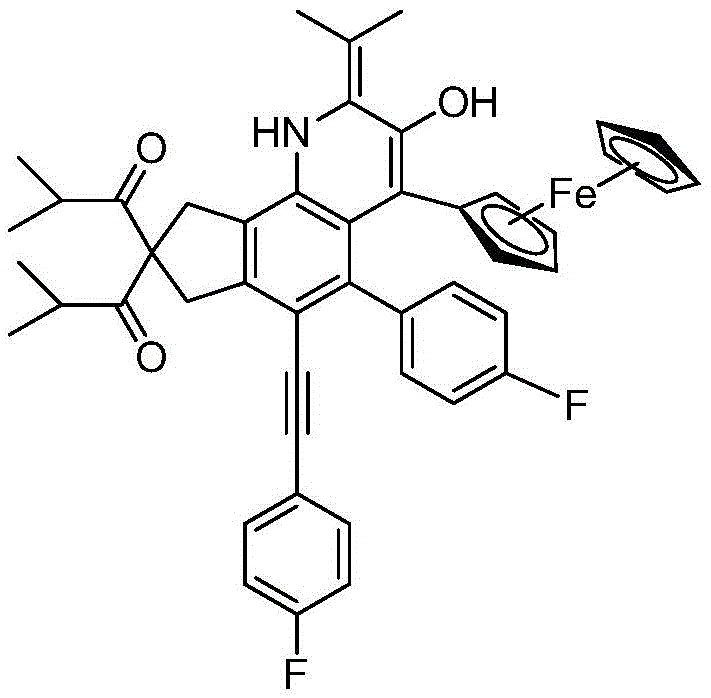
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the reduction of synthetic steps, which inherently lowers the consumption of solvents, reagents, and energy. By avoiding the isolation of unstable intermediates and utilizing a one-pot cascade strategy, the overall process mass intensity (PMI) is significantly improved compared to linear synthesis routes. The removal of expensive transition metal catalysts in the final cyclization step, relying instead on thermal activation, further decreases the bill of materials. Additionally, the simplified purification workflow, which utilizes standard crystallization techniques rather than preparative HPLC, ensures that production costs remain competitive even at large scales. These factors collectively contribute to a more economical manufacturing profile for high-value quinoline intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, operating at atmospheric pressure and moderate temperatures, reduces the risk of batch failures due to equipment malfunction or parameter deviation. The precursors used in this synthesis are derived from widely available bulk chemicals, mitigating the risk of supply chain bottlenecks associated with specialized custom synthons. The scalability of the thermal cyclization step allows for seamless technology transfer from pilot plants to full-scale commercial production facilities without the need for significant process re-engineering. This reliability ensures consistent delivery schedules for downstream customers, supporting just-in-time manufacturing models in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The use of toluene as the primary solvent aligns with established industrial safety protocols, and its recovery and recycling are well-understood processes that minimize environmental impact. The high selectivity of the reaction reduces the formation of toxic byproducts, simplifying the effluent treatment requirements and facilitating compliance with increasingly stringent environmental regulations. The solid nature of the final product allows for easy handling, packaging, and transportation, reducing the logistical complexities associated with liquid intermediates. This combination of scalability and environmental stewardship makes the technology an attractive option for companies aiming to enhance their sustainability profiles while expanding their production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoline synthesis technology. These answers are derived directly from the experimental data and mechanistic descriptions provided in the patent literature to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What is the key mechanistic advantage of this quinoline synthesis route?
A: The process utilizes a Hexadehydro-Diels-Alder (HDDA) reaction to generate a reactive benzyne intermediate in situ, which undergoes a cascade cyclization with the ferrocene auxiliary, offering superior atom economy compared to traditional stepwise constructions.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the reaction employs common solvents like toluene and operates at moderate temperatures (100-110°C), avoiding cryogenic conditions or ultra-high pressure, which facilitates straightforward scale-up from laboratory to multi-ton manufacturing.
Q: How is the stereochemistry controlled in the final quinoline product?
A: Stereochemical integrity is maintained through the use of a chiral ferrocene auxiliary, specifically (S)-(4-isopropyloxazolin-2-yl)ferrocene, which directs the nucleophilic attack and subsequent rearrangement steps to preserve chirality in the complex scaffold.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing advanced synthetic technologies to maintain a competitive edge in the global marketplace. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this quinoline synthesis method can be successfully translated into reliable supply chains. We are committed to delivering high-purity quinoline derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex organometallic cascades and thermal cyclizations positions us as a preferred partner for the development and supply of next-generation pharmaceutical intermediates.
We invite you to collaborate with us to explore the full potential of this technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this novel route for your target molecules. We encourage you to contact us today to obtain specific COA data for our reference standards and to discuss route feasibility assessments tailored to your volume requirements. Let us help you accelerate your development timeline with our proven expertise in fine chemical manufacturing.
