Scalable Production of High-Purity Tetrazole Intermediates via Magnetic Nano-Catalysis
Scalable Production of High-Purity Tetrazole Intermediates via Magnetic Nano-Catalysis
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing critical heterocyclic intermediates. A significant breakthrough in this domain is detailed in patent CN101805307B, which discloses a novel method for preparing tetrazole compounds using magnetic nano gamma-Fe2O3 as a heterogeneous catalyst. This technology represents a paradigm shift from traditional homogeneous catalysis, addressing long-standing challenges regarding catalyst recovery, product purity, and environmental impact. By utilizing cheap and easily prepared magnetic nanoparticles, this process enables the synthesis of various substituted tetrazoles with high efficiency and minimal pollution. For R&D directors and procurement managers, this patent offers a compelling value proposition: a robust synthetic route that simplifies purification and drastically reduces the reliance on expensive, toxic transition metals.
Tetrazoles are privileged structures in medicinal chemistry, often serving as bioisosteres for carboxylic acids due to their metabolic stability and lipophilicity. However, their synthesis has historically been plagued by the use of hazardous reagents and difficult-to-remove catalysts. The method described in CN101805307B utilizes benzonitrile or substituted benzonitrile and sodium azide as raw materials in dimethylformamide (DMF) solvent. The core innovation lies in the catalyst system, which not only drives the [2+3] cycloaddition effectively but also allows for seamless separation post-reaction. This dual functionality of catalysis and magnetic separability makes it an ideal candidate for a reliable tetrazole intermediate supplier looking to optimize their manufacturing portfolio.
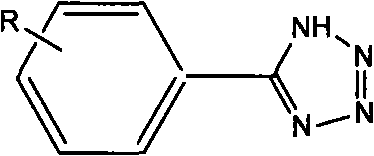
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrazole derivatives has relied heavily on Lewis acid catalysts such as zinc salts, copper salts, or other soluble transition metals. While these homogeneous catalysts often exhibit good activity, they suffer from severe drawbacks that hinder industrial application. The primary issue is the difficulty in separating the catalyst from the final product, which often necessitates complex and costly purification steps like column chromatography or extensive aqueous washing. Furthermore, the presence of residual heavy metals in the final API intermediate is a major regulatory concern, requiring stringent and expensive metal scavenging processes to meet pharmacopeial standards. Additionally, these traditional catalysts are typically single-use, generating significant amounts of heavy metal waste that complicates environmental compliance and increases disposal costs.
The Novel Approach
The novel approach presented in patent CN101805307B overcomes these limitations by employing magnetic nano gamma-Fe2O3. This heterogeneous catalyst offers a distinct advantage: it can be easily separated from the reaction mixture simply by applying an external magnetic field or through centrifugation, eliminating the need for complex filtration of fine powders. The patent data demonstrates that this catalyst can be recovered and reused for at least 10 cycles with maintained catalytic activity, which translates to substantial operational savings. The reaction conditions are also remarkably mild, typically proceeding at temperatures between 110°C and 130°C, which reduces energy consumption compared to harsher thermal methods. This methodology supports cost reduction in pharmaceutical intermediate manufacturing by streamlining the workflow and minimizing waste generation.
Mechanistic Insights into Gamma-Fe2O3 Catalyzed Cycloaddition
The mechanism of this transformation involves the activation of the nitrile group by the Lewis acidic sites on the surface of the gamma-Fe2O3 nanoparticles. The iron centers coordinate with the nitrogen atom of the nitrile, increasing the electrophilicity of the carbon atom and facilitating the nucleophilic attack by the azide ion. This surface-mediated reaction ensures that the cycloaddition proceeds efficiently to form the tetrazole ring. The magnetic nature of the catalyst does not interfere with the electronic properties required for catalysis but adds a physical dimension for separation. The use of DMF as a solvent provides a polar environment that stabilizes the ionic intermediates and dissolves the sodium azide effectively, ensuring homogeneous reaction kinetics despite the heterogeneous catalyst.
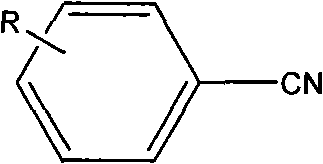
Impurity control is another critical aspect where this mechanism excels. Since the catalyst is solid and insoluble in the reaction medium under the working conditions, there is minimal leaching of iron into the solution. This results in a cleaner crude product profile with significantly lower levels of metal contaminants compared to homogeneous zinc or copper catalysis. The patent examples show consistent yields across various substrates, including electron-withdrawing groups like nitro and chloro, as well as electron-donating groups like methyl and amino. This broad substrate tolerance indicates a robust catalytic cycle that is not easily poisoned by functional groups, ensuring high-purity tetrazole compounds suitable for downstream coupling reactions in drug synthesis.
How to Synthesize Tetrazole Compounds Efficiently
The synthesis protocol outlined in the patent is straightforward and amenable to standard chemical processing equipment. It involves charging the nitrile substrate, sodium azide, and the magnetic catalyst into a reactor with DMF, followed by heating and stirring. The simplicity of the operation reduces the training burden on plant personnel and minimizes the risk of operational errors. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided below to guide process chemists in replicating this efficient route.
- Prepare the reaction mixture by combining benzonitrile or substituted benzonitrile with sodium azide in dimethylformamide (DMF) solvent.
- Add magnetic nano gamma-Fe2O3 catalyst (10-50% mass ratio) and heat the mixture to 110-130°C with stirring for 20-30 hours.
- Separate the catalyst magnetically or via centrifugation, wash with ethyl acetate for reuse, and isolate the product by pH adjustment and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this magnetic catalytic technology offers transformative benefits beyond mere chemical yield. The ability to reuse the catalyst multiple times directly impacts the bill of materials, reducing the recurring cost of catalyst procurement. Furthermore, the simplified workup procedure—eliminating complex metal scavenging and extensive washing—shortens the overall batch cycle time. This efficiency gain enhances the throughput of existing manufacturing facilities without the need for capital-intensive equipment upgrades. The process aligns perfectly with the goals of commercial scale-up of complex heterocyclic intermediates, offering a balance between performance and operational simplicity.
- Cost Reduction in Manufacturing: The elimination of expensive homogeneous catalysts and the associated purification consumables leads to a leaner cost structure. Since the gamma-Fe2O3 catalyst is inexpensive to prepare and can be recycled numerous times without significant loss of activity, the effective cost per kilogram of the catalyst is negligible. Additionally, the reduced need for water and solvents during the workup phase lowers utility costs and waste treatment expenses, contributing to a more sustainable and economically viable production model.
- Enhanced Supply Chain Reliability: The raw materials for this process, including benzonitriles and sodium azide, are commodity chemicals with stable global supply chains. The robustness of the reaction against variations in substrate electronics means that supply disruptions for specific substituted nitriles can be managed more flexibly. Moreover, the ease of catalyst recovery ensures that production is not halted waiting for fresh catalyst batches, thereby reducing lead time for high-purity tetrazole derivatives and ensuring consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling up magnetic separation is technically feasible and often simpler than scaling up filtration processes for fine catalytic powders. The process generates less hazardous waste, as the spent catalyst can be regenerated or disposed of with less environmental impact than heavy metal sludge. This aligns with increasingly stringent environmental regulations, reducing the risk of compliance-related shutdowns and enhancing the company's reputation as a green chemistry leader in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this tetrazole synthesis technology. These answers are derived directly from the experimental data and technical disclosures within patent CN101805307B, providing clarity on the feasibility and advantages of implementing this route in a commercial setting.
Q: What are the advantages of using gamma-Fe2O3 over traditional Lewis acid catalysts?
A: Unlike traditional soluble zinc or copper salts which are difficult to remove and cause heavy metal contamination, magnetic nano gamma-Fe2O3 is a heterogeneous catalyst. It can be easily separated from the reaction mixture using magnetic fields or simple centrifugation, allowing for multiple reuse cycles without significant loss of activity, thereby reducing waste and purification costs.
Q: What is the typical yield range for this tetrazole synthesis method?
A: According to the experimental data in patent CN101805307B, the method achieves high yields ranging from approximately 75% to 93% depending on the substituents on the benzonitrile ring. For instance, p-methylbenzonitrile yielded 93%, while o-chlorobenzonitrile yielded 75%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up. The use of a magnetic catalyst simplifies the downstream processing significantly compared to filtration of fine powders or complex aqueous workups required for homogeneous catalysts. The mild reaction conditions (120°C) and the stability of the catalyst over at least 10 reuse cycles support robust commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrazole Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our team of expert process chemists has extensively evaluated the magnetic nano-catalytic route described in CN101805307B and confirmed its potential for large-scale application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of tetrazole intermediate meets the highest quality standards required by top-tier drug developers.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain with high-quality, cost-effective tetrazole intermediates produced via cutting-edge green chemistry.
