Scalable Synthesis of Fully Acylated-4-Thio-D-Ribose from D-Glucose for Antiviral Intermediates
Scalable Synthesis of Fully Acylated-4-Thio-D-Ribose from D-Glucose for Antiviral Intermediates
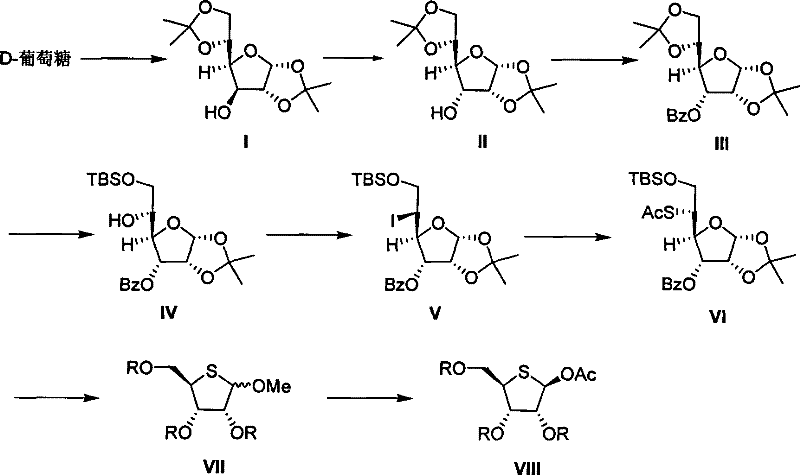
The development of efficient synthetic routes for modified nucleosides remains a critical priority in the pharmaceutical industry, particularly for the production of antiviral and antitumor agents. Patent CN101200463B discloses a groundbreaking methodology for the preparation of fully acylated-4-thio-D-ribose, a pivotal building block for 4'-thionucleosides. Unlike traditional methods that rely on scarce and expensive starting materials, this innovation utilizes D-glucose, a ubiquitous and cost-effective carbohydrate, as the primary feedstock. This strategic shift in raw material selection addresses significant economic bottlenecks in the supply chain while maintaining high stereochemical control throughout the synthesis. For R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers, this technology represents a substantial opportunity to optimize manufacturing costs without compromising on the purity required for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-thio-D-ribose and its derivatives has been heavily dependent on L-lyxose as the starting material. L-lyxose is an unnatural sugar that is not readily available in bulk quantities, leading to exorbitant raw material costs and volatile supply availability. Furthermore, existing synthetic pathways often involve complex protection and deprotection sequences that result in lower overall yields and increased waste generation. The reliance on such specialized starting materials creates a fragile supply chain, making it difficult for manufacturers to scale up production to meet the demands of commercial API manufacturing. Additionally, the purification of intermediates derived from L-lyxose can be challenging due to the presence of closely related impurities, necessitating extensive chromatographic separations that further drive up operational expenses and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The patented process introduces a robust alternative by leveraging the abundance of D-glucose. The synthetic strategy involves a series of well-defined transformations including selective protection, stereospecific iodination, and sulfur incorporation. By employing tert-butyldimethylsilyl (TBS) groups for C-6 hydroxyl protection and benzoyl groups for other positions, the route ensures high regioselectivity. The core innovation lies in the conversion of the glucose scaffold into the ribose configuration through oxidative cleavage and cyclization steps. This approach not only drastically simplifies the operation by using common reagents but also achieves a total yield of approximately 30 percent, which is highly respectable for a molecule of this complexity. For stakeholders focused on cost reduction in API manufacturing, this route offers a clear path to margin improvement through raw material substitution and process intensification.
Mechanistic Insights into Stereospecific Iodination and Thio-Substitution
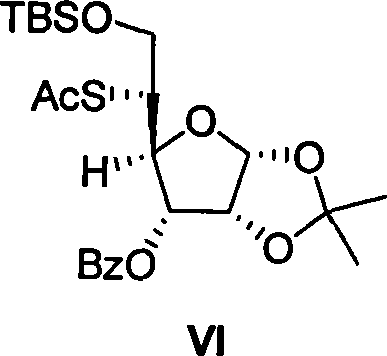
The success of this synthesis hinges on the precise control of stereochemistry during the functionalization of the furanose ring. The transformation of compound IV to compound V involves a stereospecific iodination of the C-5 hydroxyl group. Utilizing a combination of triphenylphosphine, imidazole, and iodine in an ether-acetonitrile solvent system at temperatures between 50°C and 100°C, the reaction proceeds with high efficiency to yield the 5-iodo-5-deoxy-alpha-L-talofuranoside derivative. This step is critical as it sets the stage for the subsequent nucleophilic attack. The use of mild conditions prevents the degradation of the acid-sensitive isopropylidene protecting group, ensuring the integrity of the furanose ring is maintained throughout the harsh halogenation process.
Following the iodination, the introduction of the sulfur moiety is achieved through a nucleophilic substitution reaction with potassium thioacetate. As illustrated in the reaction scheme below, compound V reacts in N,N-dimethylformamide (DMF) at elevated temperatures to produce the 5-S-acetyl-5-thio-alpha-D-allofuranoside derivative (compound VI). This step effectively inverts or retains the configuration depending on the specific mechanistic pathway (SN2), ultimately positioning the sulfur atom correctly for the formation of the thio-ribose skeleton. The subsequent steps involve deprotection of the isopropylidene group, oxidative cleavage of the resulting 1,2-diol using sodium periodate, and acidic cyclization to form the methyl thioglycoside. This sequence demonstrates a sophisticated manipulation of carbohydrate chemistry to achieve the desired 4-thio-ribose architecture from a gluco-configured precursor.
How to Synthesize Fully Acylated-4-Thio-D-Ribose Efficiently
The synthesis of this high-value intermediate requires strict adherence to reaction parameters to maximize yield and purity. The process begins with the preparation of the protected glucose derivative, followed by the critical iodination and thio-substitution steps described previously. Operators must ensure anhydrous conditions during the halogenation phase to prevent side reactions. Following the formation of the thio-intermediate, the oxidative cleavage and cyclization steps demand precise pH control and temperature management to avoid over-oxidation or degradation of the sensitive thio-glycosidic bond. The detailed standardized synthesis steps are provided in the guide below for technical teams to implement immediately.
- Stereospecific iodination of the C-5 hydroxyl group of the protected glucose derivative using triphenylphosphine and iodine to form the 5-iodo-5-deoxy intermediate.
- Nucleophilic substitution with potassium thioacetate in DMF to introduce the sulfur atom, yielding the 5-S-acetyl-5-thio derivative.
- Deprotection, oxidative cleavage of the diol, and subsequent acylation to finalize the fully acylated-4-thio-D-ribose structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to a D-glucose-based synthesis route offers transformative benefits beyond simple raw material cost savings. The stability of the glucose supply market ensures consistent pricing and availability, shielding manufacturers from the volatility associated with specialty sugars like L-lyxose. Furthermore, the simplicity of the reagents used—such as iodine, triphenylphosphine, and potassium thioacetate—means that sourcing is straightforward and does not require specialized vendor qualifications. This accessibility significantly enhances supply chain reliability and reduces the risk of production stoppages due to material shortages. The robust nature of the chemistry also allows for easier technology transfer between manufacturing sites, facilitating global production networks.
- Cost Reduction in Manufacturing: The elimination of expensive L-lyxose as a starting material results in a drastic reduction in direct material costs. Additionally, the use of common organic solvents and reagents minimizes procurement overhead. The process avoids the need for exotic catalysts or extreme conditions, which lowers energy consumption and equipment wear. By streamlining the synthetic sequence and improving the overall yield to around 30 percent, the cost per kilogram of the final intermediate is significantly optimized, allowing for more competitive pricing in the final API market.
- Enhanced Supply Chain Reliability: D-glucose is a commodity chemical produced on a massive scale globally, ensuring a secure and continuous supply. Unlike niche starting materials that may have single-source suppliers, glucose is available from multiple vendors, reducing supply risk. The intermediates generated in this process are stable and can be stored or transported with standard precautions, simplifying logistics. This reliability is crucial for maintaining uninterrupted production schedules for critical antiviral medications, ensuring that downstream customers receive their orders on time.
- Scalability and Environmental Compliance: The synthetic route is designed for scalability, utilizing reactions that are well-understood in industrial settings, such as nucleophilic substitutions and oxidative cleavages. The waste profile is manageable, with aqueous workups and standard organic extractions that fit within existing environmental treatment frameworks. The avoidance of heavy metal catalysts simplifies waste disposal and reduces the burden on effluent treatment plants. This environmental compatibility facilitates regulatory approval and supports sustainable manufacturing practices, which are increasingly important for corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4-thio-D-ribose derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these details is essential for quality assurance teams and process engineers who are evaluating the feasibility of adopting this new synthetic route for commercial production.
Q: What is the primary advantage of using D-glucose over L-lyxose for this synthesis?
A: D-glucose is an inexpensive, naturally abundant commodity chemical, whereas L-lyxose is an expensive, unnatural sugar. Switching to D-glucose significantly reduces raw material costs and improves supply chain reliability for large-scale manufacturing.
Q: What is the overall yield of the patented synthesis route?
A: According to patent CN101200463B, the total yield for the conversion of D-glucose to the final fully acylated-4-thio-D-ribose product reaches approximately 30 percent, which is competitive for a multi-step carbohydrate modification sequence.
Q: How is the sulfur atom introduced into the ribose skeleton?
A: The sulfur atom is introduced via a nucleophilic substitution reaction. A 5-iodo intermediate is reacted with potassium thioacetate in N,N-dimethylformamide (DMF) at elevated temperatures (50-100°C) to form the 5-thio derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fully Acylated-4-Thio-D-Ribose Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient nucleoside synthesis in the development of next-generation antiviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex carbohydrate chemistry allows us to offer this specific intermediate with consistent quality and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to our glucose-based supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency and technical excellence.
