Scalable Synthesis of Fully Acylated-4-Thio-D-Ribose from D-Glucose for Antiviral Drug Development
The pharmaceutical industry's relentless pursuit of potent antiviral and antitumor agents has placed 4'-thionucleosides at the forefront of medicinal chemistry research due to their exceptional metabolic stability and biological activity. Patent CN101200463B introduces a groundbreaking synthetic methodology that addresses a critical bottleneck in the production of these valuable compounds: the efficient preparation of fully acylated-4-thio-D-ribose. Historically, the synthesis of this crucial sugar scaffold relied heavily on expensive and scarce non-natural L-lyxose, creating significant supply chain vulnerabilities and cost barriers for drug developers. This patent discloses a novel, economically viable route starting from the abundant and inexpensive commodity chemical D-glucose, fundamentally altering the cost structure of producing these high-value pharmaceutical intermediates. The disclosed method not only simplifies the operational complexity but also achieves a respectable total yield, making it an attractive option for industrial adoption.
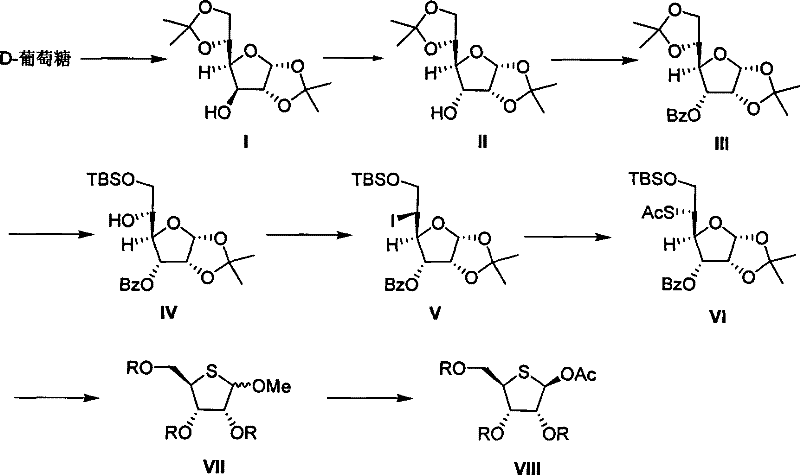
This comprehensive reaction scheme illustrates the strategic transformation of D-glucose derivatives into the target 4-thio-D-ribose scaffold. The pathway elegantly navigates through stereochemical adjustments and functional group manipulations, specifically highlighting the conversion of the C-5 hydroxyl group into a leaving group and subsequently replacing it with a sulfur moiety. By leveraging readily available starting materials and robust chemical transformations, this process offers a reliable alternative to legacy methods, ensuring a more stable supply of high-purity nucleoside building blocks for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 4-thio-D-ribose and its derivatives was predominantly dependent on L-lyxose as the starting material, a sugar that is both naturally rare and commercially expensive. Literature references such as those by E.J. Reist and R.L. Whistler highlight early attempts to construct these molecules, but they often suffered from low overall efficiency and reliance on difficult-to-source chiral pools. The scarcity of L-lyxose creates a fragile supply chain where price volatility can severely impact the economics of downstream drug production. Furthermore, traditional routes often involved complex protection and deprotection sequences that added unnecessary steps, reduced overall yields, and generated significant chemical waste. For procurement managers and supply chain heads, these factors translate into higher costs of goods sold (COGS) and increased risk of production delays due to raw material shortages.
The Novel Approach
The novel approach detailed in this patent circumvents these issues by utilizing D-glucose, one of the most abundant and cost-effective carbohydrates available globally. The strategy involves a clever sequence of reactions that effectively remodels the glucose skeleton into the desired ribose configuration with a sulfur substitution at the C-4 position. Key to this success is the preparation of specific intermediates, namely the 5-iodo-5-deoxy-alpha-L-talofuranoside derivatives (Compound V) and the 5-thio-alpha-D-allofuranoside derivatives (Compound VI). By employing tert-butyldimethylsilyl (TBS) groups for selective protection and utilizing stereospecific iodination followed by nucleophilic substitution with potassium thioacetate, the process achieves high selectivity. This methodological shift not only drastically reduces raw material costs but also simplifies the operational workflow, using common solvents like ether, acetonitrile, and DMF, which are easily sourced and handled in standard manufacturing facilities.
Mechanistic Insights into Stereospecific Iodination and Thio-Substitution
The core chemical innovation lies in the precise manipulation of the furanose ring stereochemistry to install the sulfur atom. The process begins with the activation of the C-5 hydroxyl group of the protected glucose derivative (Compound IV). Under the described conditions, typically involving triphenylphosphine and iodine in an ether-acetonitrile solvent system at elevated temperatures (50-100 degrees Celsius), a stereospecific iodination occurs. This transformation converts the primary alcohol into an iodide (Compound V) with high fidelity, setting the stage for the subsequent nucleophilic attack. The use of mild heating ensures complete conversion while minimizing side reactions that could compromise the integrity of the sensitive acetal protecting groups present on the molecule. This step is critical as it establishes the leaving group necessary for the introduction of the sulfur atom.
Following the formation of the iodo-intermediate, the synthesis proceeds with a nucleophilic substitution reaction using potassium thioacetate in N,N-dimethylformamide (DMF). This step replaces the iodine atom with a thioacetate group, yielding Compound VI. The reaction conditions, maintained between 50-100 degrees Celsius, facilitate the SN2-type displacement, effectively inverting the configuration or maintaining the desired stereochemistry required for the final ribose structure. Subsequent steps involve the removal of the isopropylidene protecting group using trifluoroacetic acid, followed by oxidative cleavage of the resulting diol with sodium periodate. This sequence shortens the carbon chain appropriately and sets up the final cyclization and acylation steps. The rigorous control over reaction parameters ensures that impurities are minimized, resulting in a product profile that meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Fully Acylated-4-Thio-D-Ribose Efficiently
The synthesis of this complex carbohydrate intermediate requires careful attention to reaction conditions and purification protocols to ensure high quality and yield. The patent outlines a robust procedure that begins with the preparation of the iodo-sugar intermediate, followed by sulfur incorporation and final deprotection. Each step is optimized to balance reaction rate with selectivity, utilizing standard workup procedures such as aqueous extraction and silica gel chromatography. For research and development teams looking to replicate or scale this process, understanding the nuances of the protection group strategy—specifically the interplay between the TBS ether and the benzoyl esters—is essential for success. The detailed experimental examples provided in the patent serve as a reliable blueprint for laboratory execution.
- Prepare 5-iodo-5-deoxy-alpha-L-talofuranoside derivatives (Compound V) from protected D-glucose precursors using stereospecific iodination.
- React Compound V with potassium thioacetate to form 5-S-acetyl-5-thio-alpha-D-allofuranoside derivatives (Compound VI).
- Perform deprotection, oxidative cleavage, and full acylation to yield the final 2,3,5-tri-O-acyl-4-thio-D-ribose product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from L-lyxose to D-glucose represents a transformative opportunity to optimize cost structures and enhance supply security. The reliance on a commodity chemical like D-glucose eliminates the risks associated with sourcing niche, high-cost sugars that are subject to market fluctuations and limited availability. This fundamental change in the bill of materials allows for significant cost reduction in pharmaceutical intermediate manufacturing without compromising on the quality or stereochemical purity of the final product. Furthermore, the use of standard reagents and solvents means that the process can be implemented in existing multipurpose chemical plants without the need for specialized equipment or hazardous handling protocols that might delay regulatory approval.
- Cost Reduction in Manufacturing: The substitution of expensive L-lyxose with inexpensive D-glucose serves as the primary driver for cost optimization in this synthesis. By removing the dependency on a scarce chiral pool starting material, manufacturers can achieve substantial savings on raw material expenditures. Additionally, the streamlined reaction sequence reduces the consumption of solvents and reagents per kilogram of product, further lowering the variable costs associated with production. The elimination of complex, low-yielding steps found in older methodologies contributes to a more efficient use of resources, directly impacting the bottom line.
- Enhanced Supply Chain Reliability: Sourcing D-glucose is inherently more stable than sourcing L-lyxose, as glucose is produced on a massive scale for the food and fermentation industries. This abundance ensures that supply disruptions are highly unlikely, providing a secure foundation for long-term production planning. The reagents used in the subsequent steps, such as triphenylphosphine, iodine, and potassium thioacetate, are also widely available industrial chemicals. This accessibility reduces lead times for high-purity pharmaceutical intermediates and allows for more flexible inventory management strategies, ensuring continuous operation even during periods of global supply chain stress.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The use of standard purification techniques like silica gel chromatography and crystallization facilitates the isolation of high-purity products consistent with Good Manufacturing Practice (GMP) standards. Moreover, the simplified synthetic route potentially reduces the generation of hazardous waste compared to more convoluted traditional methods. This alignment with green chemistry principles supports environmental compliance goals and simplifies the permitting process for new manufacturing lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of fully acylated-4-thio-D-ribose. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation. They are intended to clarify the feasibility, advantages, and specific chemical transformations involved in this novel pathway. Understanding these details is crucial for stakeholders evaluating the potential integration of this technology into their existing supply chains.
Q: What is the primary advantage of this synthesis route over conventional methods?
A: The primary advantage is the use of inexpensive D-glucose as the starting material instead of costly and unnatural L-lyxose, significantly reducing raw material costs while maintaining a viable total yield of approximately 30 percent.
Q: What are the key intermediates in this pathway?
A: The key intermediates include the 5-iodo-5-deoxy-alpha-L-talofuranoside derivative (Compound V) and the 5-S-acetyl-5-thio-alpha-D-allofuranoside derivative (Compound VI), which facilitate the introduction of the sulfur atom at the C-4 position.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes common reagents such as triphenylphosphine, iodine, and potassium thioacetate, and employs standard purification techniques like silica gel chromatography, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fully Acylated-4-Thio-D-Ribose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation antiviral therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. We are committed to delivering fully acylated-4-thio-D-ribose with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our capability to adapt complex carbohydrate chemistry to large-scale manufacturing makes us an ideal partner for your drug development programs.
We invite you to engage with our technical procurement team to discuss how this innovative D-glucose route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this supply source. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality, reliability, and technical excellence in the field of fine chemical intermediates.
