Advanced Boronate Synthesis Via NHC-Stabilized Hydroboration For Commercial Scale-Up
Advanced Boronate Synthesis Via NHC-Stabilized Hydroboration For Commercial Scale-Up
Introduction To The Patented Technology
The landscape of organoboron chemistry is undergoing a significant transformation driven by the need for safer, more stable, and highly efficient synthetic methodologies for pharmaceutical intermediates. A pivotal development in this field is documented in Chinese patent CN111718363B, which discloses a novel preparation method for boronate compounds utilizing azacarbene borane complexes. This technology addresses the longstanding challenges associated with traditional borane reagents, which are often plagued by instability and toxicity issues that complicate large-scale manufacturing processes. By leveraging a catalytic system based on tris(pentafluorophenyl)borane, the patented process enables the hydroboration of olefins under remarkably mild conditions while maintaining exceptional reaction control. For global procurement leaders and R&D directors, this represents a critical opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with reduced operational hazards. The implications of this technology extend beyond mere synthesis, offering a robust pathway for cost reduction in electronic chemical manufacturing and complex API production where boron functionality is essential.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organoboron compounds has relied heavily on the use of free borane species or unstable borane-ether complexes that pose significant safety and handling challenges in an industrial environment. These conventional reagents are frequently pyrophoric, requiring stringent inert atmosphere conditions and specialized equipment that drastically increase capital expenditure and operational complexity for chemical manufacturers. Furthermore, the lack of stability in traditional borane adducts often leads to decomposition during storage or transport, resulting in inconsistent reagent quality and unpredictable reaction outcomes that compromise the purity of the final active pharmaceutical ingredients. The necessity for rigorous safety protocols and the potential for hazardous waste generation further exacerbate the environmental burden, making these legacy methods increasingly untenable for modern sustainable chemistry initiatives. Consequently, supply chain heads face considerable risks regarding continuity and compliance when relying on these outdated synthetic routes for critical building blocks.
The Novel Approach
In stark contrast to these legacy methodologies, the approach detailed in patent CN111718363B utilizes stable Lewis base borane adducts, specifically azacarbene boranes, which offer superior stability and ease of preparation without compromising reactivity. This innovative strategy allows for the direct hydroboration of 1,2-diphenylethylene derivatives in the presence of a specific catalyst, facilitating a streamlined workflow that eliminates the need for hazardous free borane handling. The use of N-heterocyclic carbene (NHC) stabilized boranes ensures that the reagent remains intact until activated by the catalyst, providing a controlled release of reactive species that enhances both safety and selectivity. 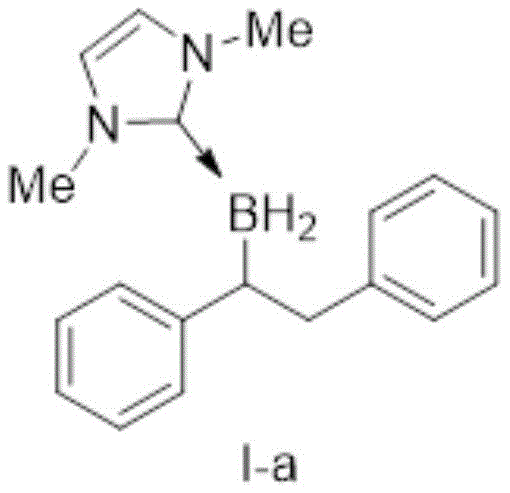 This structural stability translates directly into commercial advantages, as it simplifies logistics and reduces the regulatory burden associated with transporting dangerous goods. For a reliable agrochemical intermediate supplier or pharma partner, adopting this technology means achieving substantial cost savings through improved process safety and reduced waste disposal requirements.
This structural stability translates directly into commercial advantages, as it simplifies logistics and reduces the regulatory burden associated with transporting dangerous goods. For a reliable agrochemical intermediate supplier or pharma partner, adopting this technology means achieving substantial cost savings through improved process safety and reduced waste disposal requirements.
Mechanistic Insights into B(C6F5)3-Catalyzed Hydroboration
The core of this technological breakthrough lies in the sophisticated mechanistic interplay between the azacarbene borane and the tris(pentafluorophenyl)borane catalyst, which acts as a potent Lewis acid to activate the boron-hydrogen bond. Upon interaction with the catalyst, the NHC-borane complex undergoes a polarization that increases the electrophilicity of the boron center, thereby facilitating the nucleophilic attack by the olefin substrate in a highly regulated manner. This catalytic cycle ensures that the hydroboration proceeds with high efficiency even at moderate temperatures, typically around 80°C, which is significantly lower than many thermal hydroboration processes that require extreme conditions. 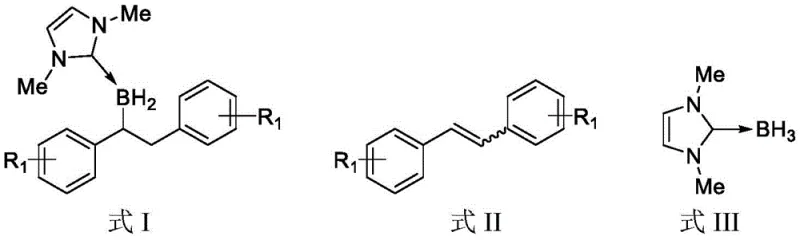 The precise control over the reaction kinetics minimizes the formation of by-products such as over-reduced species or polymerization artifacts, which are common pitfalls in uncatalyzed systems. For R&D directors focused on impurity profiles, this mechanism offers a clear advantage by delivering a cleaner crude reaction mixture that requires less intensive downstream purification, ultimately preserving yield and reducing solvent consumption during isolation.
The precise control over the reaction kinetics minimizes the formation of by-products such as over-reduced species or polymerization artifacts, which are common pitfalls in uncatalyzed systems. For R&D directors focused on impurity profiles, this mechanism offers a clear advantage by delivering a cleaner crude reaction mixture that requires less intensive downstream purification, ultimately preserving yield and reducing solvent consumption during isolation.
Furthermore, the impurity control mechanism inherent in this catalytic system is designed to accommodate a wide range of electronic and steric environments on the olefin substrate without loss of performance. The catalyst demonstrates remarkable tolerance towards various substituents, including electron-withdrawing groups like halogens and trifluoromethyl groups, as well as electron-donating alkyl and alkoxy moieties, ensuring broad applicability across diverse chemical spaces. This versatility is crucial for the commercial scale-up of complex polymer additives or specialty chemicals where substrate variability is high. The stability of the NHC ligand prevents catalyst deactivation pathways that often plague transition metal systems, ensuring consistent turnover numbers throughout the reaction duration. By maintaining a stable catalytic environment, the process guarantees batch-to-batch reproducibility, a key metric for supply chain reliability and quality assurance in regulated industries.
How to Synthesize Boronate Compound Efficiently
Implementing this synthesis route involves a straightforward procedure that begins with the charging of a reaction vessel under an inert nitrogen atmosphere to prevent moisture interference with the Lewis acid catalyst. The process requires the precise combination of the 1,2-diphenylethylene substrate and the azacarbene borane reagent in an anhydrous solvent such as toluene, followed by the addition of the catalyst in molar quantities ranging from 5% to 30%. The reaction mixture is then heated to a temperature between 60°C and 80°C for a period of 6 to 12 hours, allowing the hydroboration to proceed to completion with high conversion rates. Following the reaction, standard workup procedures involving dilution with dichloromethane and purification via column chromatography yield the target boronate compounds with excellent purity specifications suitable for downstream coupling reactions. This operational simplicity makes it an ideal candidate for technology transfer from laboratory scale to pilot plant operations.
- Prepare the reaction vessel under inert atmosphere and charge with 1,2-diphenylethylene derivative and azacarbene borane reagent.
- Add tris(pentafluorophenyl)borane catalyst and anhydrous toluene solvent to initiate the hydroboration transformation.
- Heat the mixture to 80°C for 6 hours, then purify the crude product via column chromatography to isolate the target boronate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this NHC-borane hydroboration technology offers profound benefits that directly impact the bottom line and operational resilience of chemical manufacturing organizations. The elimination of pyrophoric reagents removes the need for specialized containment infrastructure and reduces insurance premiums associated with hazardous material handling, leading to significant overhead reductions. Moreover, the high yields and selectivity reported in the patent data suggest a more efficient use of raw materials, minimizing waste generation and aligning with green chemistry principles that are increasingly mandated by global regulatory bodies. For procurement managers, this translates into a more predictable cost structure and reduced exposure to volatile pricing of specialized safety equipment. The ability to source stable reagents from commercial vendors further de-risks the supply chain, ensuring that production schedules are not disrupted by reagent degradation or availability issues.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drives cost reduction in pharmaceutical intermediate manufacturing by streamlining the synthesis workflow and eliminating expensive quenching and neutralization steps required for free boranes. The high atom economy of the hydroboration reaction ensures that the majority of the starting mass is incorporated into the final product, reducing the volume of waste solvent that requires treatment and disposal. Additionally, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys resistant to highly corrosive borane species. These factors combine to lower the overall cost of goods sold, providing a competitive edge in pricing strategies for high-value fine chemicals.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly enhanced due to the commercial availability and stability of the key reagents, specifically the azacarbene borane and the B(C6F5)3 catalyst, which can be sourced from multiple global suppliers. Unlike sensitive organometallic reagents that require cold chain logistics, these materials can be stored and transported under ambient conditions, reducing logistics costs and the risk of spoilage during transit. This robustness ensures reducing lead time for high-purity pharmaceutical intermediates, as manufacturers can maintain leaner inventory levels without fear of reagent expiration. Consequently, production planning becomes more agile, allowing for faster response times to market demands and urgent customer orders.
- Scalability and Environmental Compliance: The scalability of this process is supported by the use of common organic solvents like toluene and the absence of heavy metal catalysts, which simplifies waste stream management and environmental compliance reporting. The reaction generates minimal hazardous by-products, facilitating easier permitting for new production lines and reducing the environmental footprint of the manufacturing site. This alignment with sustainability goals is increasingly important for securing contracts with multinational corporations that have strict vendor codes of conduct regarding environmental stewardship. The process is inherently designed for commercial scale-up, capable of transitioning from kilogram to multi-ton annual commercial production with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boronate synthesis technology, derived from the specific advantages outlined in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. These insights provide a foundational understanding of why this method is superior to traditional hydroboration techniques in terms of safety, efficiency, and product quality.
Q: What are the stability advantages of NHC-stabilized boranes over traditional boranes?
A: NHC-stabilized boranes exhibit superior thermal and oxidative stability compared to traditional pyrophoric borane reagents, significantly reducing handling risks and storage requirements in industrial settings.
Q: How does the B(C6F5)3 catalyst influence reaction selectivity?
A: The Lewis acidic nature of tris(pentafluorophenyl)borane activates the borane species specifically for olefin insertion, minimizing side reactions and ensuring high regioselectivity for the desired anti-Markovnikov addition product.
Q: Is this synthesis method scalable for commercial manufacturing?
A: Yes, the use of commercially available starting materials and mild thermal conditions in common solvents like toluene facilitates straightforward scale-up from laboratory to multi-ton production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boronate Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented hydroboration technology and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to the global market. Our facility is equipped with stringent purity specifications and rigorous QC labs that ensure every batch of boronate compound meets the exacting standards demanded by the pharmaceutical and agrochemical industries. We understand that consistency is key, and our process engineering team is dedicated to optimizing these catalytic cycles to maximize yield and minimize impurity profiles for our clients. By partnering with us, you gain access to a supply chain that is not only robust but also deeply knowledgeable about the nuances of organoboron chemistry.
We invite your technical procurement team to contact us for a Customized Cost-Saving Analysis that evaluates how implementing this specific synthesis route can optimize your current manufacturing expenses. Please reach out to request specific COA data and route feasibility assessments tailored to your target molecules, allowing us to demonstrate our capability as a trusted partner in your supply chain. Our commitment to transparency and technical excellence ensures that we can support your R&D and production needs with the highest level of professionalism and reliability.
