Advanced Rh(III) Catalytic Strategy for Efficient Nitrogen Heterocycle Manufacturing
Advanced Rh(III) Catalytic Strategy for Efficient Nitrogen Heterocycle Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex nitrogen-containing heterocyclic scaffolds, which serve as critical cores for numerous bioactive molecules. A significant breakthrough in this domain is detailed in patent CN109232529B, which discloses a novel preparation method for constructing azacyclo skeletons via self-assembly guiding group-assisted Rh(III) catalysis. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot protocol that generates the effective directing group in situ. By utilizing simple aromatic aldehydes and 2-aminopyridine as starting materials, this method successfully realizes a synthetic strategy that combines high atom economy with operational simplicity. For R&D directors and procurement managers alike, this innovation offers a compelling route to access high-purity isoindolinone frameworks and isoquinoline structures with substitution characteristics, addressing key challenges in modern API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of isoindolinone skeletons via C-H activation has relied heavily on pre-functionalized substrates, which introduces significant inefficiencies into the supply chain. Early methodologies, such as those reported by Li Xingwei's group in 2010, utilized N-phenyl-substituted arylformamides as substrates to react with acrylates. While these methods established the feasibility of Heck-type intermediates leading to cyclization, they suffered from severe limitations regarding substrate scope and reaction severity. Specifically, when the aromatic ring contained heterocycles such as furan or indole, the reaction often stalled at the oxidation Heck product stage rather than proceeding to the desired isoindolinone. Furthermore, subsequent improvements by Yu Jinquan's group in 2014, which employed N-perfluoro-substituted phenyl arylcarboxamides, although milder, required complex and expensive raw materials. These conventional approaches necessitate the separate synthesis of the amide directing group prior to the cyclization step, adding extra unit operations, increasing waste generation, and driving up the overall cost of goods sold for the final pharmaceutical intermediate.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN109232529B introduces a transformative one-pot strategy that eliminates the need for pre-synthesized amide substrates. This novel approach leverages the condensation of readily available aromatic aldehydes with 2-aminopyridine to generate an efficient directing group in situ within the reaction vessel. This self-assembled system then undergoes Rh(III)-catalyzed C-H activation and subsequent reaction with substituted olefins to yield the target nitrogen heterocyclic skeleton. The reaction conditions are remarkably mild, typically operating at 80°C in acetonitrile, which significantly reduces energy consumption compared to high-temperature alternatives. As illustrated in the general reaction scheme below, this method accommodates a wide variety of substituents on both the aromatic aldehyde and the olefin, demonstrating exceptional versatility.
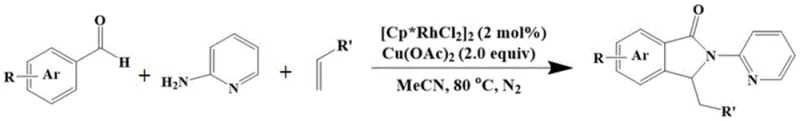
The ability to start from simpler raw materials not only simplifies the logistical burden on the supply chain but also enhances the overall atom economy of the process. By merging the directing group formation and the cyclization into a single operational sequence, manufacturers can drastically reduce solvent usage, labor hours, and purification steps, thereby achieving substantial cost reduction in pharmaceutical intermediate manufacturing without compromising on yield or purity.
Mechanistic Insights into Rh(III)-Catalyzed C-H Activation and Cyclization
For technical stakeholders evaluating the robustness of this synthesis, understanding the underlying catalytic cycle is paramount. The mechanism initiates with the condensation of the aromatic aldehyde and 2-aminopyridine to form an imine species, which acts as the transient directing group. This species coordinates with the Rh(III) catalyst, facilitating the critical C-H activation step at the ortho-position of the aromatic ring. Following activation, the substituted olefin undergoes migratory insertion into the Rh-C bond. The subsequent steps involve cyclization and beta-hydride elimination or reductive elimination processes that restore the aromaticity and release the final isoindolinone product while regenerating the active catalyst species. The detailed catalytic cycle, depicted below, highlights the elegance of this self-assembly strategy where the directing group is both formed and consumed within the catalytic manifold.
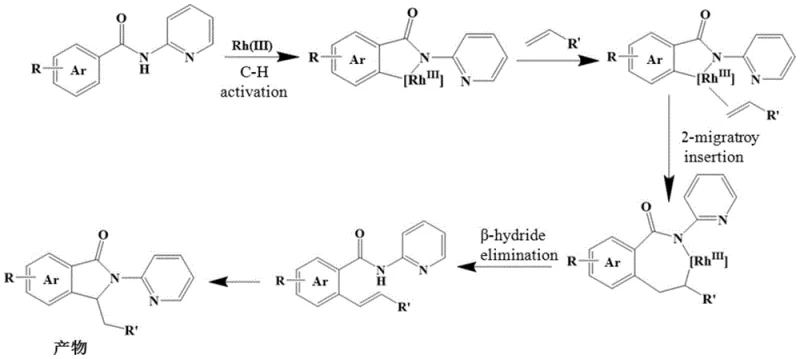
From an impurity control perspective, the mild reaction temperature of 80°C plays a crucial role in minimizing side reactions such as polymerization of the olefin or over-oxidation of the sensitive aldehyde starting material. The use of copper acetate as a stoichiometric oxidant ensures efficient turnover of the rhodium catalyst while maintaining a clean reaction profile. This mechanistic clarity allows process chemists to fine-tune parameters such as the electronic nature of the substituents (R and R') to optimize reaction rates and selectivity, ensuring that the final product meets the stringent purity specifications required for downstream drug substance synthesis.
How to Synthesize Isoindolinone Derivatives Efficiently
Implementing this advanced Rh(III) catalytic protocol requires precise attention to reagent quality and atmospheric control to maximize yield and reproducibility. The standard procedure involves charging a Schlenk bottle with the aromatic aldehyde, 2-aminopyridine, copper acetate, and the rhodium dimer catalyst under an inert atmosphere. After establishing a nitrogen environment through vacuum-nitrogen cycles, the solvent and substituted olefin are introduced, and the mixture is heated to facilitate the tandem condensation and cyclization sequence. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated in the patent examples, are provided in the guide below.
- Combine aromatic aldehyde, 2-aminopyridine, copper acetate oxidant, and [Cp*RhCl2]2 catalyst in a Schlenk bottle under inert atmosphere.
- Add polar solvent such as acetonitrile and the substituted olefin substrate to the reaction mixture.
- Heat the mixture to 80°C for 4-12 hours, then purify the resulting isoindolinone product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this self-assembly Rh(III) catalytic method offers tangible strategic benefits that extend beyond mere technical novelty. The primary value proposition lies in the drastic simplification of the raw material portfolio. By replacing complex, custom-synthesized amide substrates with commodity chemicals like aromatic aldehydes and 2-aminopyridine, companies can mitigate supply risks associated with specialized intermediates. This shift not only stabilizes the supply chain but also leverages the economies of scale inherent in bulk chemical markets, leading to significant cost optimization. Furthermore, the one-pot nature of the reaction reduces the number of isolation and purification stages, which directly translates to lower manufacturing overheads and reduced waste disposal costs, aligning perfectly with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of the pre-synthesis step for the directing group removes an entire unit operation from the production schedule, effectively cutting labor and utility costs associated with that stage. Additionally, the high atom economy ensures that a greater proportion of the input mass is converted into the valuable product, minimizing raw material waste. The use of a robust catalytic system that operates at moderate temperatures further reduces energy expenditures compared to high-pressure or high-temperature alternatives, contributing to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key starting materials, aromatic aldehydes and aminopyridines, are widely available from multiple global suppliers, reducing dependency on single-source vendors. This abundance ensures consistent availability and price stability, shielding the production schedule from the volatility often seen with niche fine chemical intermediates. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, enhancing overall batch-to-batch consistency and reliability.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common solvents like acetonitrile make this process highly amenable to scale-up from laboratory to commercial production volumes. The simplified workflow reduces the generation of hazardous waste streams, facilitating easier compliance with environmental regulations. Moreover, the high yields reported in the patent examples suggest that the process is efficient enough to support large-scale commercial production of complex pharmaceutical intermediates without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Rh(III) catalytic technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for decision-making. Understanding these details is essential for assessing the feasibility of integrating this method into existing production lines or new product development pipelines.
Q: What is the primary advantage of this Rh(III) catalytic method over conventional approaches?
A: The primary advantage is the in-situ generation of the directing group from simple aromatic aldehydes and 2-aminopyridine, eliminating the need for pre-synthesizing complex amide substrates and significantly improving atom economy.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction operates under mild conditions, typically at 80°C in acetonitrile solvent, using 2 mol% [Cp*RhCl2]2 catalyst and 2 equivalents of copper acetate as the oxidant under a nitrogen atmosphere.
Q: Is this methodology suitable for large-scale commercial production?
A: Yes, the process utilizes readily available starting materials, avoids harsh reaction conditions, and employs a simple one-pot procedure, making it highly scalable and compliant with green chemistry principles for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Rh(III)-mediated self-assembly strategy for producing high-value nitrogen heterocycles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isoindolinone intermediate delivered meets the highest industry standards for pharmaceutical applications.
We invite you to leverage our technical expertise to optimize your supply chain and reduce time-to-market for your critical drug candidates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about adopting this efficient and sustainable manufacturing solution for your next-generation therapeutics.
