Revolutionizing Chiral Isoindolinone Production with Advanced Rhodium Catalysis for Commercial Scale
Revolutionizing Chiral Isoindolinone Production with Advanced Rhodium Catalysis for Commercial Scale
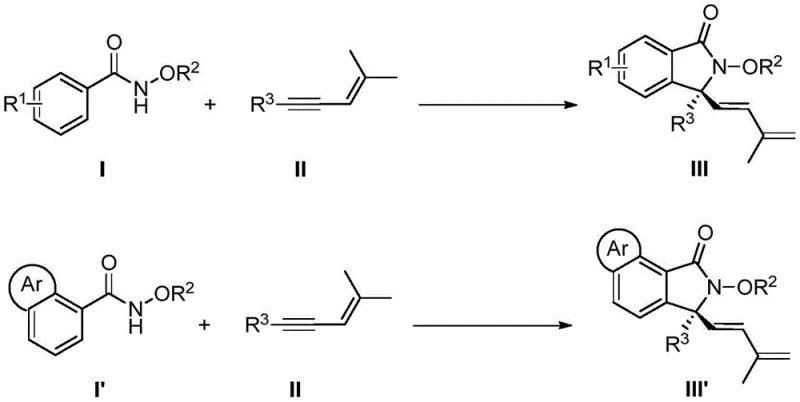
The pharmaceutical industry constantly seeks more efficient pathways to construct complex chiral scaffolds, particularly those found in bioactive molecules. Patent CN113735756A introduces a groundbreaking methodology for the synthesis of chiral 3,3-disubstituted isoindolinone compounds, a privileged structural motif prevalent in numerous drug candidates. This innovation leverages a specifically designed chiral cyclopentadienyl rhodium catalyst to facilitate a tandem sequence involving C-H bond activation, enyne migration insertion, 1,4-rhodium migration, and nucleophilic cyclization. Unlike traditional approaches that often suffer from harsh conditions and poor selectivity, this protocol operates under remarkably mild temperatures ranging from 5°C to 15°C. The ability to construct quaternary carbon centers with high enantioselectivity directly from simple N-methoxybenzamides and 1,3-enynes represents a significant leap forward in synthetic efficiency. For R&D teams focused on process optimization, this technology offers a robust platform for generating diverse libraries of chiral intermediates with minimal environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3,3-disubstituted chiral isoindolinone skeletons has been a formidable challenge for organic chemists due to the steric congestion around the quaternary carbon center. Conventional strategies typically rely on the enantioselective functionalization of pre-existing isoindolinone cores, a route that is inherently limited by the availability and stability of the starting materials. These traditional methods often necessitate multi-step synthetic sequences involving protection and deprotection groups, which drastically reduce the overall atom economy and increase the generation of chemical waste. Furthermore, the requirement for harsh reaction conditions or expensive chiral auxiliaries often leads to scalability issues, making it difficult to transition from milligram-scale discovery to kilogram-scale production. The low step economy not only inflates the cost of goods but also extends the lead time for process development, creating bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct C-H activation strategy that bypasses the need for pre-functionalized substrates, effectively streamlining the synthetic route into a single catalytic step. By employing a chiral cyclopentadienyl rhodium catalyst, the reaction achieves a formal [4+1] cyclization between N-methoxybenzamides and 1,3-enynes, constructing the target skeleton with exceptional precision. This approach transforms simple, commercially available building blocks into high-value chiral products without the burden of extensive substrate preparation. The mild reaction conditions, specifically the low temperature range, minimize side reactions such as polymerization or decomposition, which are common pitfalls in transition metal catalysis. This paradigm shift not only enhances the sustainability of the manufacturing process but also provides a versatile platform capable of accommodating a wide range of substituents, thereby expanding the chemical space accessible for drug discovery programs.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
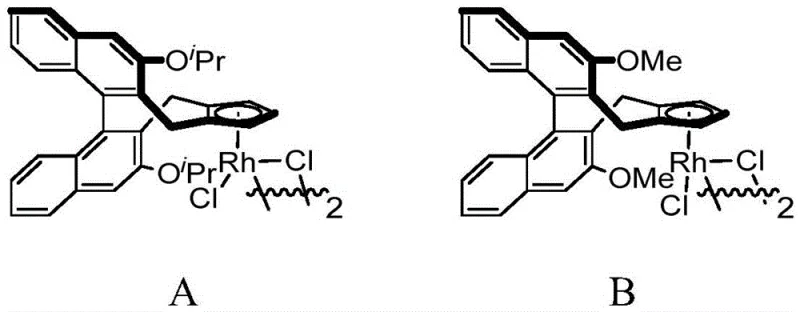
The success of this transformation hinges on the unique properties of the chiral cyclopentadienyl rhodium catalyst, which orchestrates a complex series of elementary steps with high fidelity. The catalytic cycle initiates with the coordination of the rhodium species to the directing group of the N-methoxybenzamide, facilitating the cleavage of the ortho C-H bond to form a stable rhodacycle intermediate. This C-H activation step is crucial as it sets the stage for the subsequent insertion of the 1,3-enyne substrate. The unique electronic and steric environment provided by the chiral ligand ensures that the enyne inserts in a specific orientation, which is the primary determinant of the stereochemical outcome of the reaction. Following insertion, a rare 1,4-rhodium migration occurs, shifting the metal center to a position that enables the final nucleophilic attack by the amide oxygen.
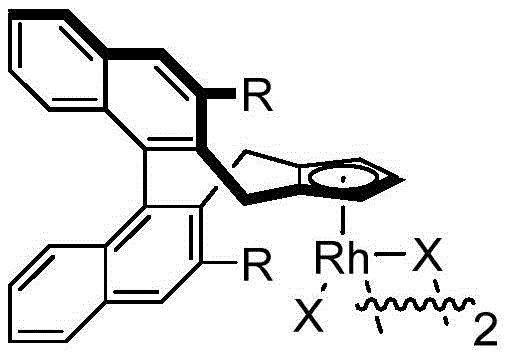
This intricate dance of bond formation and migration is tightly controlled by the chiral pocket of the catalyst, preventing the formation of racemic byproducts. The use of silver difluoride as an oxidant plays a pivotal role in regenerating the active Rh(III) species from the reduced Rh(I) state, closing the catalytic loop efficiently. Understanding this mechanism is vital for process chemists aiming to troubleshoot potential impurities; for instance, deviations in temperature or oxidant stoichiometry could disrupt the delicate balance of the migration steps, leading to regioisomers. The robustness of this catalytic system allows for the tolerance of various functional groups, including halogens, esters, and ethers, without compromising the integrity of the chiral center. This mechanistic clarity provides a solid foundation for scaling the reaction, as the rate-determining steps are well-defined and manageable under standard industrial reactor conditions.
How to Synthesize Chiral 3,3-Disubstituted Isoindolinone Efficiently
Implementing this synthesis requires careful attention to reagent quality and reaction parameters to maximize yield and enantiomeric purity. The protocol is designed to be operationally simple, utilizing standard laboratory equipment and readily available solvents such as 3-pentanol or ethanol. The key to success lies in the precise control of the reaction temperature, which must be maintained strictly between 5°C and 15°C throughout the 60 to 80-hour duration to ensure optimal catalyst performance. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining N-methoxybenzamide, 1,3-eneyne compound, chiral cyclopentadienyl rhodium catalyst, oxidant, and carboxylic acid additive in an alcohol solvent.
- Maintain the reaction temperature between 5-15°C and stir continuously for a duration of 60 to 80 hours to ensure complete conversion.
- Quench the reaction with ethylenediamine, remove solvents, and purify the crude product via silica gel column chromatography to isolate the target chiral compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials; since the method utilizes stable and commodity-grade N-methoxybenzamides and enynes, there is no reliance on exotic or custom-synthesized precursors that often carry long lead times and high price volatility. This shift to common feedstocks significantly de-risks the sourcing strategy, ensuring a more resilient supply chain capable of withstanding market fluctuations. Furthermore, the high atom economy of the [4+1] cyclization means that less raw material is wasted as byproduct, directly correlating to a reduction in the cost of goods sold (COGS) and a smaller environmental footprint.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences and the use of low catalyst loadings (3-5 mol%) substantially lower the operational expenses associated with labor, energy, and waste disposal. By avoiding the need for cryogenic conditions or high-pressure reactors, the process reduces capital expenditure requirements for specialized equipment, allowing for production in existing general-purpose facilities. The high yields reported, often exceeding 80-90%, mean that less starting material is required per unit of output, further driving down the variable costs of production. Additionally, the simplified purification process, typically requiring only standard silica gel chromatography, reduces the consumption of expensive solvents and stationary phases.
- Enhanced Supply Chain Reliability: The stability of the reactants and the mild nature of the reaction conditions contribute to a highly reliable manufacturing process with consistent batch-to-batch reproducibility. This reliability is critical for maintaining continuous supply to downstream API manufacturers, minimizing the risk of stockouts or delays that can disrupt drug development timelines. The broad substrate scope demonstrated in the patent implies that a single production line can be adapted to synthesize a wide variety of analogues, providing flexibility to respond quickly to changing market demands or clinical trial requirements. This adaptability ensures that the supply chain remains agile and responsive to the dynamic needs of the pharmaceutical sector.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic profile being manageable under mild temperatures, reducing the safety risks associated with large-scale exothermic reactions. The use of alcohol solvents and the absence of toxic heavy metal residues in the final product simplify the regulatory compliance landscape, facilitating faster approval for GMP manufacturing. The high enantioselectivity reduces the need for costly and wasteful chiral resolution steps, aligning with green chemistry principles and corporate sustainability goals. This combination of safety, scalability, and environmental stewardship makes the technology an attractive option for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this Rh-catalyzed method over traditional synthesis?
A: This method utilizes direct C-H bond activation, eliminating the need for pre-functionalized substrates and multi-step sequences, thereby significantly improving atom economy and reducing waste generation compared to conventional routes.
Q: What level of enantioselectivity can be achieved with this process?
A: The process demonstrates excellent stereocontrol, achieving enantiomeric excess (ee) values up to 95% and yields reaching 91%, ensuring the production of high-purity chiral intermediates suitable for pharmaceutical applications.
Q: Is this synthesis scalable for industrial manufacturing?
A: Yes, the reaction operates under mild conditions (5-15°C) using stable and commercially available raw materials, which simplifies thermal management and safety protocols, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this rhodium-catalyzed cyclization can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, to guarantee the quality of every batch delivered. We understand that consistency and reliability are paramount in the pharmaceutical supply chain, and our dedicated technical team is committed to optimizing these complex syntheses for maximum efficiency and cost-effectiveness.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions about your supply chain strategy. Let us be your partner in turning complex chemical challenges into commercial successes.
