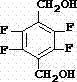
Advanced Synthesis of 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol for High-Purity Pyrethroid Production
The global demand for high-performance pyrethroid insecticides continues to drive the need for efficient synthesis of key intermediates like 2,3,5,6-tetrafluoro-1,4-benzenedimethanol. Patent CN103641686A introduces a groundbreaking synthetic methodology that addresses critical bottlenecks in traditional manufacturing, specifically targeting the production of this vital agrochemical intermediate. The disclosed technology shifts away from costly and hazardous reagents, such as methyl lithium, towards a more sustainable and economically viable pathway starting from 2,3,5,6-tetrachloro-1,4-terephthalonitrile. This innovation is particularly significant for manufacturers seeking to optimize their supply chains for products like tefluthrin and perfluthrin. By integrating catalyst-free fluorination and a novel one-step alcoholysis process, the patent outlines a route that not only enhances product purity but also drastically simplifies the operational workflow. For R&D directors and procurement specialists, understanding these mechanistic improvements is essential for evaluating long-term sourcing strategies and ensuring consistent quality in the final pesticide formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,5,6-tetrafluoro-1,4-benzenedimethanol has been plagued by inefficient multi-step processes that rely on expensive and dangerous reagents. One prominent conventional route utilizes hexafluorobenzene as the starting material, requiring methylation with methyl lithium, followed by bromination and reduction. This approach is fundamentally flawed for large-scale production due to the exorbitant cost of organolithium reagents and the inherent safety risks associated with handling them on an industrial scale. Furthermore, the yield across these multiple steps is often suboptimal, leading to significant material loss and increased waste generation. Another traditional pathway involves starting from terephthalic acid, which necessitates chlorination and fluorination steps that frequently result in complex mixtures of by-products. These side reactions complicate purification efforts and lower the overall economic efficiency of the process. Additionally, older nitrile-based routes often involve separate hydrolysis and esterification steps, which prolong the production cycle and increase the consumption of solvents and energy, thereby inflating the cost of goods sold for the final agrochemical intermediate.
The Novel Approach
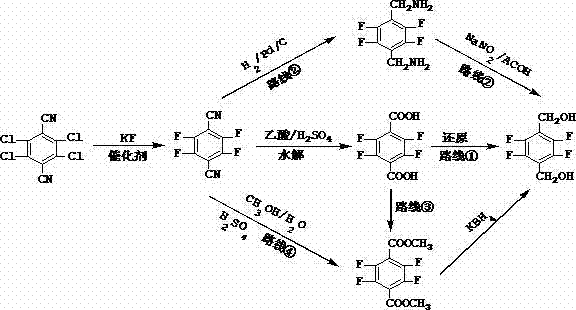
The innovative methodology presented in the patent data offers a transformative solution by streamlining the synthesis into a concise and robust sequence. Starting from 2,3,5,6-tetrachloro-1,4-terephthalonitrile, the process employs a direct fluorination with potassium fluoride in DMF, notably eliminating the need for phase transfer catalysts in this initial step to reduce impurity formation. A key breakthrough is the one-step alcoholysis reaction, where the nitrile group is directly converted to a dimethyl ester using methanol and sulfuric acid, bypassing the traditional isolation of the carboxylic acid intermediate. This consolidation of steps not only saves time but also allows for the recycling of the acidic mother liquor, significantly mitigating environmental impact. The final reduction utilizes potassium borohydride in an aqueous system with a phase transfer catalyst, replacing flammable organic solvents like ethanol. This holistic approach ensures higher yields, superior product quality, and a much safer operating environment, making it the preferred choice for modern chemical manufacturing.
Mechanistic Insights into Catalyst-Free Fluorination and Aqueous Reduction
The core of this technological advancement lies in the precise control of reaction conditions to maximize selectivity and minimize side reactions. In the initial fluorination step, the reaction between 2,3,5,6-tetrachloro-1,4-terephthalonitrile and potassium fluoride is conducted in DMF at elevated temperatures ranging from 110°C to 120°C. Unlike previous methods that relied on phase transfer catalysts which could introduce colored impurities and complicate downstream purification, this novel protocol operates effectively without them. The absence of these additives results in a product that is off-white rather than reddish-brown, indicating a cleaner reaction profile with fewer polymeric or oxidized by-products. This purity is critical for subsequent steps, as impurities carried forward can poison catalysts or interfere with crystallization. The nucleophilic aromatic substitution proceeds efficiently due to the strong electron-withdrawing nature of the nitrile groups, which activate the ring for fluoride attack, ensuring high conversion rates without the need for excessive reagent loading.
Furthermore, the reduction mechanism represents a significant departure from standard practices by utilizing water as the primary solvent medium. Traditionally, the reduction of esters to alcohols using borohydrides is performed in alcoholic solvents like ethanol or methanol to ensure solubility and reactivity. However, this patent demonstrates that by employing a quaternary ammonium salt as a phase transfer catalyst, the reduction can proceed efficiently in an aqueous environment. This shift not only reduces the fire hazard associated with large volumes of organic solvents but also simplifies the work-up procedure. The phase transfer agent facilitates the transport of the borohydride anion to the organic ester interface, enabling rapid hydride transfer. Following the reaction, simple acidification and filtration allow for the isolation of the crude product, which is then purified via recrystallization from a methanol-water mixture. This green chemistry approach aligns with modern regulatory standards while maintaining high chemical fidelity.
How to Synthesize 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol Efficiently
The implementation of this synthesis requires careful attention to temperature control and reagent stoichiometry to replicate the high yields reported in the patent literature. The process begins with the fluorination of the tetrachloro precursor, followed immediately by the one-pot alcoholysis which consolidates what were previously two distinct unit operations. This integration is crucial for maintaining throughput and minimizing solvent usage. The subsequent reduction step must be managed to control gas evolution, ensuring safe pressure relief within the reactor. Detailed standard operating procedures regarding the specific molar ratios of potassium fluoride to substrate, as well as the concentration of sulfuric acid for alcoholysis, are critical for success. For a comprehensive guide on the exact experimental parameters, including stirring rates and cooling profiles, please refer to the standardized synthesis steps outlined below.
- React 2,3,5,6-tetrachloro-1,4-terephthalonitrile with potassium fluoride in DMF at 110-120°C without a phase transfer catalyst.
- Perform one-step alcoholysis using methanol/water and sulfuric acid with a manganese dioxide catalyst to form the dimethyl ester.
- Reduce the ester using potassium borohydride in water with a phase transfer catalyst, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers substantial strategic benefits for procurement managers and supply chain directors looking to optimize their sourcing of agrochemical intermediates. The elimination of expensive reagents like methyl lithium and the reduction in the number of processing steps directly translate to a lower cost of production. By consolidating the hydrolysis and esterification into a single alcoholysis step, manufacturers can significantly reduce utility consumption and labor costs associated with additional filtration and drying operations. Moreover, the ability to recycle the sulfuric acid mother liquor means that raw material consumption is further minimized, creating a more circular and cost-effective production loop. These efficiencies allow suppliers to offer more competitive pricing structures without compromising on the quality or purity specifications required by downstream pesticide formulators.
- Cost Reduction in Manufacturing: The transition to a catalyst-free fluorination system and the use of aqueous reduction media removes the dependency on costly organic solvents and specialized catalysts. This shift drastically lowers the variable costs associated with raw material procurement and waste disposal. By avoiding the use of ethanol and ethyl acetate in the reduction phase, the process eliminates the need for complex solvent recovery systems and reduces the risk of fire-related insurance premiums. Additionally, the higher yield obtained through this optimized pathway means that less starting material is required to produce the same amount of final product, effectively stretching the value of every kilogram of input. These cumulative savings contribute to a significantly improved margin profile for the manufactured intermediate.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the risk of production delays caused by equipment failures or complex purification bottlenecks. The robustness of the one-step alcoholysis and the mild conditions of the aqueous reduction ensure that the process is less susceptible to minor fluctuations in operating parameters. This stability leads to more predictable lead times and consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to global agrochemical clients. Furthermore, the use of widely available and stable reagents like potassium fluoride and potassium borohydride mitigates the risk of supply disruptions that might occur with more exotic or hazardous chemicals. This reliability strengthens the overall resilience of the supply chain against market volatility.
- Scalability and Environmental Compliance: The design of this process prioritizes scalability and environmental stewardship, addressing two major concerns for modern chemical enterprises. The reduction in equipment corrosion, achieved by avoiding high-temperature hydrolysis with glacial acetic acid, extends the lifespan of reactors and piping, thereby lowering capital expenditure on maintenance and replacement. From an environmental standpoint, the minimization of waste acid discharge and the avoidance of volatile organic compounds (VOCs) facilitate easier compliance with stringent environmental regulations. The aqueous work-up and recycling protocols demonstrate a commitment to green chemistry principles, making the facility more attractive to investors and regulators alike. This alignment with sustainability goals ensures long-term operational viability in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2,3,5,6-tetrafluoro-1,4-benzenedimethanol. These insights are derived directly from the technical specifications and comparative data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions regarding process adoption and quality assurance. The answers reflect the specific advantages of the new methodology over legacy processes, focusing on yield, purity, and operational safety.
Q: What are the primary advantages of the novel synthesis route over conventional methods?
A: The novel route eliminates the need for expensive methyl lithium and avoids harsh hydrolysis conditions, significantly reducing equipment corrosion and waste acid discharge while improving overall yield.
Q: How does the new process improve environmental compliance?
A: By utilizing water as the solvent for the reduction step instead of ethanol or ethyl acetate and recycling sulfuric acid mother liquor, the process minimizes volatile organic compound emissions and hazardous waste.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process features mild reaction conditions, simplified unit operations, and reduced equipment corrosion, making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy of final agrochemical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,3,5,6-tetrafluoro-1,4-benzenedimethanol meets the highest industry standards. Our commitment to process optimization allows us to deliver products that are not only pure but also cost-effective, supporting your downstream manufacturing goals.
We invite you to collaborate with us to explore how this advanced synthesis technology can benefit your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current supply chain structure. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as your trusted partner in fine chemical manufacturing. Let us help you secure a reliable supply of this essential intermediate while optimizing your overall production costs.
