Scalable Photocatalytic Synthesis of Nilaparib Intermediates for Commercial API Production
Scalable Photocatalytic Synthesis of Nilaparib Intermediates for Commercial API Production
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing complex oncology drugs like Nilaparib, a potent PARP inhibitor approved for treating ovarian cancer. Patent CN113637002B introduces a groundbreaking preparation method that fundamentally shifts the synthetic paradigm from traditional hazardous chemistry to a greener, photocatalytic approach. This innovation addresses critical bottlenecks in the supply chain by replacing dangerous reagents such as sodium azide with benign alternatives driven by visible light energy. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates while adhering to increasingly strict environmental regulations. The disclosed methodology not only simplifies the reaction sequence but also enhances the overall safety profile of the production facility, making it an attractive candidate for technology transfer and commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
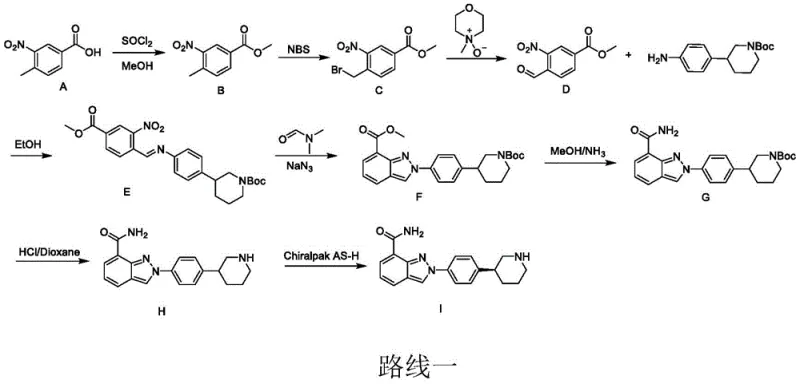
The Limitations of Conventional Methods
Historically, the synthesis of Nilaparib has been plagued by lengthy reaction sequences and the reliance on highly toxic or expensive reagents that pose significant challenges for industrial application. As illustrated in the prior art routes, traditional methods often involve up to eight distinct steps, starting from materials like 3-methyl-2-nitrobenzoic acid, which inherently limits overall yield and throughput. A major concern for supply chain managers is the use of sodium azide, a hazardous explosive substance that requires specialized handling protocols and creates substantial waste disposal issues. Furthermore, alternative routes utilizing platinum dioxide for pyridine ring reduction introduce prohibitive costs due to the precious metal catalyst, while chiral resolution steps in early stages can lead to significant material loss. These factors collectively result in a fragile supply chain with high production costs and elevated safety risks, necessitating a robust alternative for reliable pharmaceutical intermediate supplier operations.
The Novel Approach
The novel synthetic strategy disclosed in the patent offers a streamlined and economically viable alternative by leveraging modern photocatalytic techniques to construct key molecular scaffolds efficiently. Instead of relying on thermal activation or dangerous azide chemistry, this method employs a palladium-catalyzed coupling reaction driven by visible light irradiation to generate critical intermediates under mild conditions. The process begins with the coupling of compound 1 and bromobenzene, facilitated by a Pd catalyst and potassium formate, to directly yield the key piperidine derivative intermediate with high selectivity. This approach drastically reduces the number of purification steps and eliminates the need for high-pressure hydrogenation equipment, thereby lowering capital expenditure for manufacturing facilities. By integrating chiral resolution at a strategic point and utilizing copper-catalyzed coupling for the final indazole assembly, the route ensures both stereochemical purity and operational simplicity, positioning it as a superior choice for cost reduction in API manufacturing.
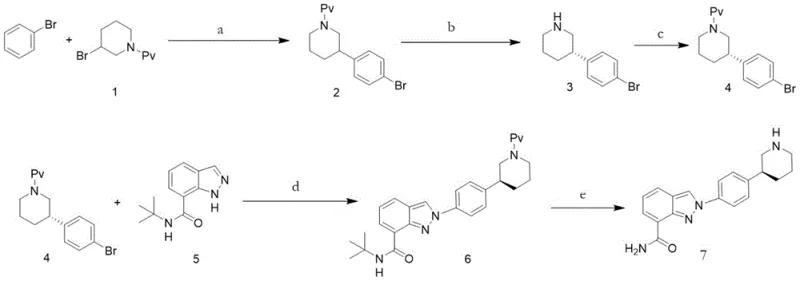
Mechanistic Insights into Pd-Catalyzed Photoredox Coupling
The core innovation of this synthesis lies in the mechanistic elegance of the palladium-catalyzed photoredox reaction, which utilizes light energy to drive single electron transfer processes that are otherwise difficult to achieve thermally. When the reaction mixture containing the Pd catalyst and substrate is irradiated with visible light (wavelengths between 450nm and 560nm), the photons excite the catalyst to a higher energy state, facilitating the generation of alkyl free radicals. These reactive species undergo addition reactions with the substrate to form the carbon-carbon bonds necessary for the piperidine-phenyl scaffold, a crucial structural motif in Nilaparib. This mechanism operates effectively at temperatures ranging from 20°C to 60°C, avoiding the thermal degradation of sensitive functional groups that often occurs in traditional high-temperature couplings. The use of potassium formate as a hydrogen source further enhances the atom economy of the reaction, ensuring that the process remains environmentally benign while maintaining high conversion rates as monitored by HPLC analysis.
Impurity control is another critical aspect where this mechanistic approach offers distinct advantages over conventional nitro-reduction or azide-based cyclizations. The mild reaction conditions minimize the formation of side products typically associated with harsh acidic or basic environments, leading to a cleaner crude reaction profile. Following the photocatalytic step, the intermediate can be purified through straightforward recrystallization or silica gel chromatography, yielding a white solid with a sharp melting point range indicative of high purity. Subsequent steps involving chiral resolution using tartaric acid derivatives and copper-mediated coupling are designed to preserve this purity, ensuring that the final active pharmaceutical ingredient meets stringent regulatory specifications. This robust control over the impurity profile is essential for R&D teams aiming to accelerate clinical development timelines by reducing the burden of extensive impurity characterization studies.
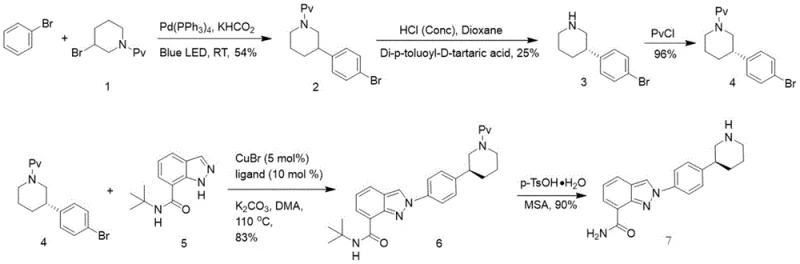
How to Synthesize Nilaparib Key Intermediate Efficiently
The synthesis of the key intermediate, 1-[3-(4-bromophenyl)piperidin-1-yl]-2,2-dimethylpropan-1-one, serves as the foundation for the entire Nilaparib production process and can be achieved with remarkable efficiency using the disclosed protocol. The procedure involves dissolving the starting amine and palladium catalyst in bromobenzene, followed by the addition of potassium formate to create a homogeneous reaction system ready for irradiation. Under visible light exposure, the reaction proceeds to completion within 24 hours, after which standard workup procedures involving extraction and concentration yield the desired product in high purity. This streamlined operation eliminates the need for complex inert atmosphere setups or cryogenic conditions, making it accessible for standard laboratory and pilot plant environments. For detailed standardized synthesis steps and specific parameter optimization, please refer to the guide below.
- Dissolve compound 1 and a Pd catalyst (e.g., Pd(PPh3)4) in bromobenzene, add potassium formate, and mix to form an orange turbid solution.
- Irradiate the solution with visible light (450nm-560nm) at 20-60°C until HPLC confirms complete consumption of compound 1.
- Perform post-treatment by removing solvent, followed by recrystallization or silica gel chromatography to isolate the key intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic synthesis route translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of hazardous reagents like sodium azide removes a significant logistical burden, as there is no longer a need for specialized storage, transport, or disposal protocols associated with explosive materials. This simplification of the chemical inventory directly contributes to enhanced supply chain reliability, ensuring that production schedules are not disrupted by regulatory hurdles or safety incidents. Furthermore, the replacement of expensive platinum catalysts with more abundant palladium systems, coupled with the use of visible light as a clean energy source, drives down the raw material and utility costs associated with manufacturing. These factors collectively create a more resilient and cost-effective supply chain capable of meeting the growing global demand for oncology therapeutics without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The transition to a photocatalytic process significantly lowers operational expenses by removing the dependency on high-cost precious metal catalysts like platinum dioxide and eliminating the need for high-pressure hydrogenation equipment. The use of visible light as the primary energy source reduces electricity consumption compared to thermal heating methods, while the simplified workup procedures minimize solvent usage and waste treatment costs. Additionally, the higher selectivity of the reaction reduces the loss of valuable starting materials, improving the overall mass balance and economic efficiency of the production line. These cumulative effects result in a substantially lower cost of goods sold, providing a competitive edge in the pricing of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: By sourcing readily available and stable reagents such as bromobenzene and potassium formate, the new method mitigates the risk of supply disruptions often caused by the scarcity of specialized chemicals. The absence of hazardous azides simplifies vendor qualification and logistics, allowing for faster procurement cycles and reduced lead times for raw material delivery. Moreover, the robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production failures that could delay shipments to downstream API manufacturers. This stability is crucial for maintaining continuous supply to pharmaceutical partners who rely on just-in-time delivery models for their clinical and commercial programs.
- Scalability and Environmental Compliance: The mild operating conditions of the photocatalytic reaction facilitate easy scale-up from laboratory to commercial production without the engineering challenges associated with high-pressure or high-temperature processes. The green chemistry principles embedded in this route, such as high atom utilization and reduced waste generation, align perfectly with global environmental regulations, minimizing the carbon footprint of the manufacturing process. This compliance not only avoids potential fines and regulatory penalties but also enhances the corporate sustainability profile, which is increasingly important for stakeholders and investors. Consequently, the process is well-suited for large-scale implementation, ensuring a steady and sustainable supply of high-quality intermediates for the pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Nilaparib synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a clear understanding of the method's capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their own manufacturing operations or for procurement professionals assessing the long-term viability of the supply source. The answers reflect a commitment to transparency and technical accuracy, ensuring that all stakeholders have the necessary information to make informed decisions.
Q: What are the safety advantages of this new Nilaparib synthesis route?
A: The new route eliminates the use of hazardous sodium azide (NaN3) and expensive platinum dioxide (PtO2) required in conventional methods, significantly reducing operational risks and environmental impact.
Q: How does the photocatalytic step improve process efficiency?
A: By utilizing visible light to excite the Pd catalyst for single electron transfer, the reaction proceeds under mild temperatures (20-60°C) without high pressure, simplifying equipment requirements and energy consumption.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the process avoids extreme conditions and uses readily available reagents like potassium formate and bromobenzene, making it highly scalable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nilaparib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology in reshaping the landscape of oncology drug manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates. We are committed to delivering high-purity Nilaparib intermediates that adhere to the highest quality standards, supporting our partners in bringing life-saving therapies to patients faster and more efficiently.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge synthesis route for their projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall manufacturing costs. Together, we can drive the next generation of pharmaceutical innovation through sustainable and efficient chemical synthesis.
