Advanced Pd-Catalyzed Synthesis of Atropisomeric 1-Arylisoquinoline N-Oxides for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex chiral scaffolds, particularly those serving as privileged structures in asymmetric catalysis. Patent CN112574107B, published in April 2023, introduces a groundbreaking methodology for the synthesis of atropisomeric 1-arylisoquinoline N-oxide derivatives. These compounds are not merely academic curiosities; they function as potent organocatalysts in critical transformations such as the asymmetric allylation of aromatic aldehydes. The core innovation lies in bypassing traditional multi-step sequences in favor of a direct, transition metal-catalyzed carbon-hydrogen bond functionalization strategy. By leveraging the intrinsic directing capability of the N-oxide moiety, this technology enables the direct construction of the sterically hindered biaryl axis in a single operational step. This represents a paradigm shift from stepwise assembly to convergent synthesis, offering profound implications for process chemistry and supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
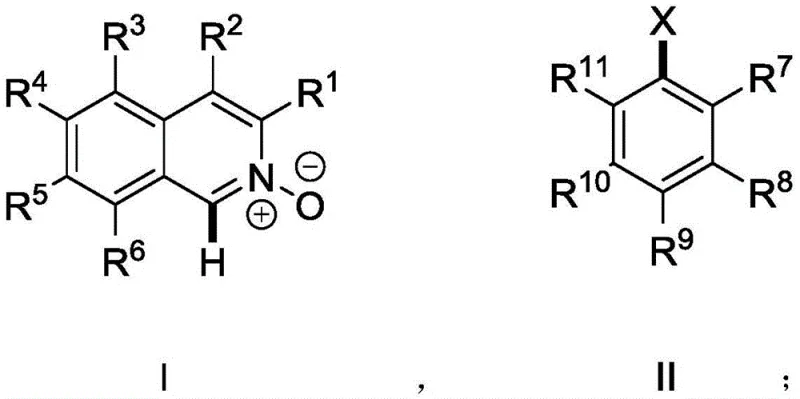
Historically, the preparation of 1-arylisoquinoline N-oxides with defined atropisomerism has been plagued by inefficiencies inherent to classical cross-coupling logic. The standard protocol, as illustrated in prior art such as the work by Malkov et al., necessitates a laborious three-stage sequence. Initially, the isoquinoline core must be chlorinated to generate a reactive 1-chloroisoquinoline intermediate. Subsequently, this chloride undergoes a Suzuki-Miyaura type coupling with an aryl boronic acid, a reagent class known for its high cost and variable stability upon storage. Finally, a separate oxidation step is required to install the crucial N-oxide functionality. This linear progression suffers from cumulative yield losses at each stage and generates significant chemical waste. Furthermore, the reliance on boronic acids introduces supply chain vulnerabilities, as these reagents are often more expensive and less commercially ubiquitous than their halide counterparts. The atom economy of such a route is inherently poor due to the stoichiometric generation of boron and halide byproducts.
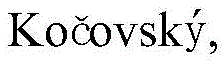
The Novel Approach
In stark contrast, the methodology disclosed in CN112574107B collapses these multiple operations into a single, elegant transformation. By employing a palladium catalyst system in the presence of a suitable base and phosphine ligand, the process achieves direct C-H arylation of the isoquinoline N-oxide scaffold. This approach eliminates the need for pre-functionalization of the heterocycle (i.e., no chlorination step) and replaces expensive boronic acids with inexpensive and widely available aryl halides. The N-oxide oxygen atom acts as an internal directing group, coordinating to the palladium center and facilitating the activation of the proximal C-H bond at the C1 position. This intramolecular assistance ensures high regioselectivity, preventing the formation of unwanted isomers that would complicate downstream purification. The result is a streamlined process that drastically reduces reaction time, solvent consumption, and overall material costs, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Pd-Catalyzed Direct C-H Arylation
The success of this transformation hinges on the delicate interplay between the palladium catalyst, the ligand environment, and the directing group effects of the N-oxide. Mechanistically, the cycle initiates with the coordination of the palladium species to the nitrogen-oxygen bond of the isoquinoline N-oxide. This coordination brings the metal center into close proximity with the C1-H bond, lowering the activation energy required for cleavage. Through a concerted metalation-deprotonation (CMD) pathway facilitated by the carbonate base, the C-H bond is activated to form a stable palladacycle intermediate. This step is critical as it defines the regioselectivity of the reaction. Following C-H activation, the aryl halide undergoes oxidative addition to the palladium center. The subsequent reductive elimination step forge the new carbon-carbon bond between the isoquinoline C1 position and the aryl ring, releasing the product and regenerating the active catalyst. The steric bulk introduced by the ortho-substituents on the aryl ring (R7 group) is what locks the rotation around the newly formed biaryl bond, thereby generating the stable atropisomeric configuration observed at room temperature.
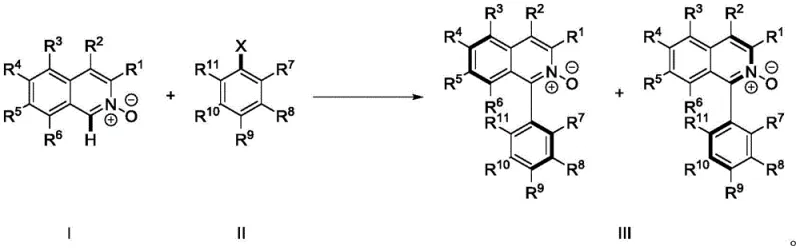
Impurity control in this system is managed through the high specificity of the N-O directed mechanism. Unlike non-directed C-H activation which can lead to mixtures of regioisomers, the chelation effect ensures that functionalization occurs almost exclusively at the C1 position. Furthermore, the choice of ligand, specifically bulky electron-rich phosphines like di-tert-butylmethylphosphine tetrafluoroborate, plays a pivotal role in suppressing homocoupling of the aryl halide, a common side reaction in palladium catalysis. The reaction conditions, typically ranging from 60°C to 140°C in solvents like toluene, are optimized to balance reaction rate with thermal stability of the N-oxide moiety. Post-reaction, the removal of palladium residues is straightforward, often achievable through standard filtration over diatomaceous earth or silica gel, ensuring the final product meets stringent purity specifications required for catalytic applications without the need for complex scavenging resins.
How to Synthesize Atropisomeric 1-Arylisoquinoline N-Oxide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters regarding catalyst loading and atmospheric control. The protocol dictates the use of an inert atmosphere, typically nitrogen, to prevent oxidation of the phosphine ligand and the palladium catalyst, which could lead to premature deactivation. The molar ratio of the isoquinoline N-oxide to the aryl halide is generally maintained at 1:3 to drive the equilibrium towards completion, while the catalyst loading is kept low, often between 5 to 15 mol%, to ensure economic viability. The base, preferably cesium carbonate, serves dual roles in neutralizing the acid byproduct and assisting in the C-H cleavage step. Detailed standard operating procedures regarding temperature ramping, quenching, and chromatographic separation are essential for reproducibility.
- Mix isoquinoline N-oxide derivative, aryl halide, palladium catalyst (e.g., Pd(OAc)2), phosphine ligand, base (e.g., Cs2CO3), and solvent (e.g., toluene) in a closed vessel under nitrogen.
- Heat the reaction mixture to 60-140°C and stir for 1-24 hours to facilitate the direct C-H bond arylation.
- Perform post-treatment including filtration through diatomaceous earth, solvent removal, and column chromatography to isolate the pure atropisomeric product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits beyond mere technical elegance. The shift from boronic acid-based coupling to aryl halide-based direct arylation fundamentally alters the cost structure of the raw material basket. Aryl halides are commodity chemicals produced on a massive scale for various industries, ensuring consistent availability and pricing stability compared to the more niche boronic acids. This substitution alone drives a significant reduction in the Bill of Materials (BOM) cost. Moreover, the consolidation of three synthetic steps into one drastically reduces the man-hours required for production, lowers utility consumption for heating and cooling across multiple stages, and minimizes the volume of solvents needed for intermediate isolations. These factors collectively contribute to a leaner manufacturing process with a smaller environmental footprint.
- Cost Reduction in Manufacturing: The elimination of the chlorination and oxidation steps removes the need for specific reagents like POCl3 and mCPBA, which can be hazardous and costly to handle on a large scale. By avoiding the isolation of the 1-chloroisoquinoline intermediate, the process saves on filtration, drying, and storage costs associated with semi-finished goods. The high yields reported in the patent examples, often exceeding 80-90%, mean that less starting material is wasted, directly improving the cost-per-kilogram of the final active pharmaceutical ingredient (API) intermediate or catalyst. This efficiency allows for more competitive pricing models when supplying downstream customers who are sensitive to margin pressures.
- Enhanced Supply Chain Reliability: Relying on a single-step conversion reduces the complexity of the production schedule. In a multi-step process, a delay or quality issue in an early step cascades through the entire timeline, jeopardizing delivery commitments. With this streamlined approach, the lead time from raw material intake to finished goods is significantly compressed. Additionally, the robustness of the reaction conditions (tolerating various substituents like methyl, methoxy, and halogens) means that the same production line can be easily adapted to synthesize a library of different derivatives without extensive re-validation, providing flexibility to meet diverse customer demands rapidly.
- Scalability and Environmental Compliance: The use of common solvents like toluene and the absence of heavy metal waste streams (beyond the recoverable palladium catalyst) simplify waste treatment protocols. The atom economy is superior because the hydrogen atom from the C-H bond and the halogen from the aryl halide are the primary byproducts, forming simple salts with the base. This reduces the burden on wastewater treatment facilities and aligns with increasingly strict global environmental regulations. The simplicity of the workup, involving basic filtration and distillation, makes the process highly amenable to scale-up from gram-scale R&D to multi-ton commercial production without encountering the engineering bottlenecks typical of complex multi-step syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and reliability for potential partners.
Q: What are the primary limitations of traditional synthesis methods for these compounds?
A: Conventional routes typically require multi-step sequences involving the initial synthesis of 1-chloroisoquinoline followed by coupling with expensive aryl boronic acids and a final oxidation step, resulting in poor atom economy and lower overall yields.
Q: How does the new Pd-catalyzed method improve raw material availability?
A: The novel approach utilizes readily available aryl halides instead of costly aryl boronic acids, significantly simplifying the supply chain and reducing the dependency on specialized coupling reagents.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method features a simple one-step reaction profile with high selectivity and yield, using common solvents like toluene and standard purification techniques, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atropisomeric 1-Arylisoquinoline N-Oxide Supplier
As the demand for chiral organocatalysts and specialized pharmaceutical intermediates continues to grow, having a manufacturing partner with deep technical expertise is crucial. NINGBO INNO PHARMCHEM stands at the forefront of process development, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of atropisomeric 1-arylisoquinoline N-oxide meets the exacting standards required for asymmetric synthesis applications. We understand that consistency is key in catalytic applications, where minor impurities can drastically affect enantioselectivity in downstream reactions.
We invite R&D directors and procurement specialists to engage with our technical team to discuss how this innovative synthesis route can be integrated into your supply chain. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your production of high-value chiral intermediates.
