Scalable Visible Light Catalyzed Dichlorination for High-Purity Pharmaceutical Intermediates
Introduction to Green Dichlorination Technology
The synthesis of dichloride-containing compounds represents a critical challenge in modern organic synthesis, particularly for the production of high-value pharmaceutical intermediates and agrochemical precursors. Patent CN111253254A introduces a groundbreaking preparation method for visible light-catalyzed dichloride addition products of aliphatic olefins, addressing significant limitations in current industrial practices. This technology leverages the unique properties of transition metal chlorides with visible light absorption capacity to initiate light-induced chlorine atom transfer, thereby facilitating efficient addition reactions under remarkably mild conditions. By utilizing visible light as the energy source and hydrochloric acid as the chlorine source, this innovation eliminates the need for harsh thermal conditions and toxic chlorinating agents that have historically plagued this sector. The process operates in an aerobic atmosphere, ensuring a sustainable and environmentally friendly workflow that aligns with modern green chemistry principles. For R&D directors and process chemists, this patent offers a robust pathway to access complex dichlorinated scaffolds with high purity and structural integrity, essential for downstream drug development.
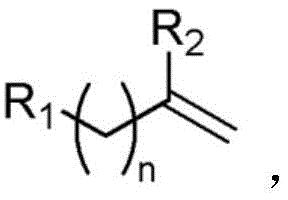
Furthermore, the versatility of this method allows for the functionalization of a wide range of aliphatic olefins, including those with terminal double bonds and various aromatic substituents. The ability to introduce two chlorine atoms regioselectively across a carbon-carbon double bond without compromising sensitive functional groups is a significant advancement. This capability is particularly valuable for the synthesis of chiral catalysts and bioactive molecules where the introduction of chlorine atoms can drastically alter physical and chemical properties to enhance biological activity. As a reliable pharmaceutical intermediate supplier, understanding such technological shifts is crucial for maintaining a competitive edge in the global market. The integration of photocatalysis into large-scale manufacturing processes signifies a move towards more energy-efficient and cost-effective production models, reducing the overall carbon footprint of chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-chlorine compounds has relied on methodologies that are fraught with significant operational and safety drawbacks. Traditional approaches often necessitate the use of expensive and highly specialized chiral catalysts, such as (DHQD)2PHAL combined with DCDMH, which limit the substrate scope primarily to allyl amide compounds while excluding common aromatic and aliphatic olefins. Other established methods involve the generation of chloronium ions using dangerous inorganic salt oxidants like potassium peroxymonosulfate and ammonium chloride, posing severe safety hazards and creating substantial environmental pollution due to heavy metal waste and toxic byproducts. Additionally, processes utilizing triphosgene and pyridine to generate chloronium-like intermediates require cumbersome pre-synthesis of epoxy compounds and operate under dangerous reaction conditions that demand rigorous safety protocols. Electrochemical methods employing transition metal manganese and lithium perchlorate electrolytes introduce further complexity and safety risks associated with strong oxidants. These conventional thermal reactions typically require high activation energies, necessitating elevated temperatures that can degrade sensitive substrates and lead to lower yields and poor selectivity.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a visible light-catalyzed strategy that fundamentally transforms the reaction landscape. By employing copper chloride as a photocatalyst, the system harnesses visible light energy to drive the reaction at room temperature, eliminating the need for external heating and significantly reducing energy consumption. This method directly utilizes commercial hydrochloric acid as the chlorine source, bypassing the need for pre-treatment or the use of hazardous chlorinating reagents like triphosgene. The reaction proceeds smoothly in an aerobic atmosphere, simplifying the setup by removing the requirement for inert gas protection often needed in sensitive organometallic catalysis. The broad substrate tolerance allows for the efficient conversion of various aliphatic olefins, including those with electron-withdrawing and electron-donating groups, into high-purity dichloride addition products. This shift from thermal to photochemical activation not only enhances safety and environmental compliance but also opens new avenues for synthesizing compounds that are inaccessible via traditional thermochemical pathways.
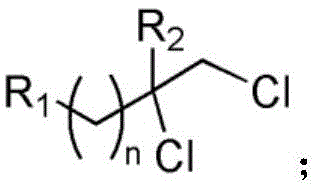
Mechanistic Insights into CuCl2-Catalyzed Photoredox Dichlorination
The core of this innovative synthesis lies in the Ligand-to-Metal Charge Transfer (LMCT) mechanism facilitated by the copper chloride catalyst under visible light irradiation. Upon absorption of photons from the 38W white LED source, the CuCl2 complex undergoes excitation, leading to the homolytic cleavage of the Cu-Cl bond and the generation of highly reactive chlorine radicals. These chlorine radicals then engage in a radical addition process with the carbon-carbon double bond of the aliphatic olefin substrate, forming a carbon-centered radical intermediate. This intermediate subsequently captures a second chlorine atom, likely from another equivalent of the activated copper species or through a radical chain propagation mechanism, to yield the final dichloride product. Crucially, the presence of oxygen in the reaction atmosphere plays a vital role in the catalytic cycle by facilitating the re-oxidation of the reduced copper species back to its active Cu(II) state, thus completing the catalytic loop and ensuring sustained turnover without the accumulation of inactive catalyst forms. This mechanistic pathway avoids the high-energy transition states associated with ionic chlorination, allowing for milder conditions and better functional group compatibility.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways which often suffer from rearrangement side reactions or over-chlorination. The specificity of the radical addition to the double bond minimizes the formation of regioisomers and poly-chlorinated byproducts, resulting in a cleaner crude reaction profile. The use of acetonitrile as the solvent further stabilizes the radical intermediates and ensures good solubility for both the organic substrates and the inorganic catalyst. Detailed analysis of the reaction outcomes, such as the conversion of substrate 1a to product 2a, confirms the high fidelity of this mechanism, with spectroscopic data matching theoretical predictions perfectly. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as light intensity and oxygen flow, to maximize efficiency and minimize waste, ensuring consistent quality for commercial scale-up of complex pharmaceutical intermediates.

How to Synthesize Dichloride Addition Products Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls to ensure optimal yield and purity. The standard protocol involves charging a reaction vessel with the aliphatic olefin substrate, 20 mol% of CuCl2 catalyst, and 2.5 equivalents of aqueous HCl in acetonitrile. The mixture is then subjected to irradiation from a 38W white LED lamp while stirring at ambient temperature (23-25°C) for a duration of 72 hours under an air atmosphere. Following the reaction, the mixture is quenched with saturated sodium sulfite to neutralize residual oxidants, extracted with ethyl acetate, and purified via column chromatography. This streamlined procedure eliminates complex workup steps and reduces solvent usage compared to traditional methods.
- Prepare the reaction mixture by combining the aliphatic olefin substrate, 20 mol% CuCl2 catalyst, and 2.5 equivalents of aqueous HCl in acetonitrile solvent.
- Irradiate the reaction system with 38W white LEDs while stirring at room temperature (23-25°C) in an aerobic atmosphere for 72 hours.
- Quench the reaction with saturated sodium sulfite solution, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light-catalyzed technology presents compelling economic and logistical benefits that directly impact the bottom line. The substitution of expensive noble metal catalysts with abundant copper salts results in a drastic reduction in raw material costs, making the production of dichlorinated intermediates significantly more affordable. Furthermore, the elimination of hazardous reagents like triphosgene and strong oxidants simplifies regulatory compliance and reduces the costs associated with waste disposal and safety management. The mild reaction conditions, operating at room temperature without the need for cryogenic cooling or high-pressure equipment, lower the energy demands of the manufacturing process, contributing to substantial cost savings in utility consumption. These factors combined enhance the overall cost-effectiveness of the supply chain, allowing for more competitive pricing strategies in the global market.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts such as iridium or ruthenium with inexpensive copper chloride significantly lowers the catalyst cost per kilogram of product. Additionally, the use of commercial hydrochloric acid instead of specialized chlorinating agents reduces reagent expenses. The simplified purification process, often requiring only basic column chromatography, minimizes solvent consumption and labor hours, leading to a leaner manufacturing cost structure. This economic efficiency is critical for maintaining margins in the highly competitive pharmaceutical intermediate sector.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents like CuCl2 and HCl ensures a robust supply chain that is less susceptible to disruptions compared to processes requiring custom-synthesized or scarce reagents. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or equipment malfunction, ensuring consistent delivery schedules. Moreover, the scalability of the photochemical setup allows for flexible production volumes, enabling rapid response to fluctuating market demands without the need for extensive capital investment in specialized high-pressure reactors.
- Scalability and Environmental Compliance: The green nature of this process, characterized by the use of visible light and non-toxic reagents, aligns perfectly with increasingly stringent environmental regulations. The absence of heavy metal waste and hazardous byproducts simplifies the permitting process for new manufacturing facilities and reduces the liability associated with environmental contamination. The energy-efficient nature of LED-driven reactions supports corporate sustainability goals, making the supply chain more resilient to future carbon taxes or energy restrictions. This forward-looking approach ensures long-term viability and market access for the produced intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light-catalyzed dichlorination technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners and technical stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines.
Q: What are the safety advantages of this dichlorination method compared to traditional chlorination?
A: Unlike traditional methods that require hazardous reagents like triphosgene or strong oxidants like potassium peroxymonosulfate, this patented process utilizes commercially available hydrochloric acid and copper chloride under mild visible light conditions, significantly reducing operational risks and environmental toxicity.
Q: Does this photocatalytic process require expensive noble metal catalysts?
A: No, the process specifically utilizes copper chloride (CuCl2), a base metal salt, as the photocatalyst. This avoids the high costs associated with precious metal catalysts like iridium, ruthenium, or palladium often found in other photoredox systems, leading to substantial raw material cost optimization.
Q: What is the substrate scope for this visible light catalyzed reaction?
A: The method demonstrates broad applicability across various aliphatic olefins containing C9-C15 chains linked to carbon-carbon double bonds, including derivatives with phthalimide, naphthyl, and substituted phenyl groups, making it versatile for synthesizing diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dichloride Addition Product Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the visible light-catalyzed dichlorination technology described in patent CN111253254A for the production of high-purity pharmaceutical intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry innovation can be realized on an industrial scale. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering consistent quality and reliability, leveraging our technical expertise to optimize reaction parameters for maximum yield and minimal impurity profiles.
We invite you to collaborate with us to explore the commercial viability of this cutting-edge synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this technology can reduce your overall manufacturing expenses. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these critical dichlorinated building blocks for your drug development pipeline.
