Advanced Chiral Resolution and Peptide Coupling Strategies for High-Purity Ubenimex Manufacturing
Advanced Chiral Resolution and Peptide Coupling Strategies for High-Purity Ubenimex Manufacturing
The global demand for high-quality antineoplastic agents continues to surge, placing immense pressure on the supply chains of critical pharmaceutical intermediates. Among these, Ubenimex stands out as a potent immunomodulator and aminopeptidase inhibitor, yet its complex stereochemistry has historically posed significant manufacturing challenges. Patent CN101891647A, filed in late 2010, introduces a transformative methodology that addresses the longstanding issues of optical purity and process scalability. This technical disclosure outlines a robust synthetic route that leverages naturally occurring amino acids as resolving agents and advanced peptide coupling reagents to achieve unprecedented purity levels. For R&D directors and procurement specialists, understanding this shift from traditional chiral amines to bio-based resolving agents represents a pivotal opportunity for cost reduction in pharmaceutical manufacturing while ensuring the stringent quality standards required for oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
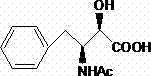
Historically, the industrial synthesis of Ubenimex has relied heavily on resolution strategies utilizing S-(-)-Alpha-Methyl benzylamine. While this approach provided a foundational route, it suffers from inherent thermodynamic limitations that prevent the complete separation of the desired (2S,3R) enantiomer from its (2R,3S) counterpart. The physical properties of these diastereomeric salts are often too similar, leading to co-crystallization and the carryover of the unwanted isomer into downstream processes. Furthermore, the subsequent peptide bond formation typically employed DCC (N,N'-dicyclohexylcarbodiimide) and HOBt, conditions known to induce partial racemization at the C-5 chiral center of the leucine residue. This results in a final product contaminated with pharmacologically inactive stereoisomers, such as N-[(2R,3S)-4-phenyl-3-amino-2-hydroxybutyryl]-L-leucine, which not only dilutes potency but complicates regulatory approval due to impurity profiling requirements.
The Novel Approach
The innovative strategy detailed in the patent data replaces expensive and less efficient chiral amines with readily available, cost-effective basic amino acids such as L-lysine, L-arginine, or L-histidine. These resolving agents exhibit superior selectivity during the salification process, preferentially forming stable salts with the target (2S,3R)-3-acetylamino-2-hydroxy-4-phenylbutyric acid while leaving the undesired enantiomer in the mother liquor. This breakthrough allows for the isolation of the key intermediate with an enantiomeric excess (ee) exceeding 99.5%, a benchmark difficult to achieve with conventional methods. Additionally, the process integrates EDCI (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) coupled with HOAt (1-hydroxy-7-azabenzotriazole) for the critical amide condensation step. This specific reagent combination drastically suppresses racemization, ensuring the stereochemical integrity of the final peptide bond is maintained throughout the synthesis.
Mechanistic Insights into Amino Acid-Mediated Chiral Resolution and Peptide Coupling
The core of this technological advancement lies in the precise molecular recognition between the racemic substrate and the chiral amino acid resolving agent. When L-lysine is introduced to the racemic mixture of (2S,3R) and (2R,3S) isomers in a optimized solvent system (such as ethanol/acetonitrile), distinct solubility differences arise between the resulting diastereomeric salts. The (2S,3R)-lysine salt possesses a crystal lattice energy that favors precipitation under controlled cooling conditions, whereas the (2R,3S) salt remains solvated. This selective crystallization is further enhanced by a crystal transformation step in a secondary solvent like isobutyl acetate, which promotes Ostwald ripening and excludes impurities from the growing crystal face. The result is a highly purified intermediate that serves as a pristine foundation for the subsequent deprotection and coupling reactions, effectively breaking the cycle of impurity propagation seen in older synthetic routes.
In the latter stages of synthesis, the preservation of chirality during peptide bond formation is paramount. Traditional carbodiimide activation often proceeds via an oxazolone intermediate, which is prone to racemization through enolization. The inclusion of HOAt acts as a powerful nucleophilic catalyst that intercepts the O-acylisourea intermediate to form an active ester with significantly lower racemization potential. The electron-withdrawing nitrogen atom at the 7-position of the azabenzotriazole ring enhances the electrophilicity of the carbonyl carbon while stabilizing the transition state, thereby accelerating the aminolysis by the leucine derivative. This mechanistic nuance ensures that the C-5 stereocenter of the leucine moiety remains untouched, preventing the formation of the inactive D-leucine isomers that plague conventional batches.
How to Synthesize (2S,3R)-3-Amino-2-hydroxy-4-phenylbutyric Acid Efficiently
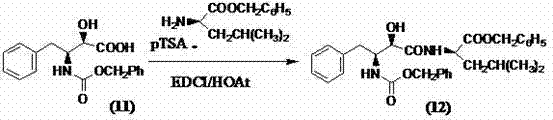
The execution of this synthesis requires careful control of solvent composition, temperature gradients, and stoichiometric ratios to maximize the efficiency of the resolution step. The process begins with the dissolution of the racemic acetyl-protected precursor in a heated alcohol-based solvent, followed by the addition of the resolving agent. Precise thermal management during the cooling phase is critical to induce nucleation of the desired salt without trapping impurities. Following isolation, the salt is converted to the free acid and subsequently deacetylated to reveal the primary amine functionality required for the final coupling. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide process engineers in replicating this high-fidelity route.
- Resolve racemic (2S,3R)/(2R,3S)-3-acetylamino-2-hydroxy-4-phenylbutyric acid using L-lysine, L-arginine, or L-histidine in a mixed solvent system to isolate the high-purity (2S,3R) isomer.
- Deprotect the acetyl group under acidic hydrolysis conditions to generate the key chiral amine intermediate (2S,3R)-3-amino-2-hydroxy-4-phenylbutyric acid.
- Perform peptide coupling with L-leucine benzyl ester using EDCI and HOAt as condensing agents to prevent racemization at the C-5 position, followed by catalytic hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this amino acid-based resolution technology offers substantial strategic benefits beyond mere technical superiority. The reliance on commodity chemicals like L-lysine and L-arginine, which are produced on a massive scale for the food and feed industries, decouples the supply chain from the volatility associated with specialized chiral reagents. This shift not only stabilizes raw material availability but also introduces a significant degree of flexibility in sourcing, allowing manufacturers to leverage competitive pricing from multiple global suppliers. Furthermore, the simplification of the purification workflow reduces the number of recrystallization cycles required, directly translating to shorter batch cycle times and increased throughput capacity without the need for additional capital investment in equipment.
- Cost Reduction in Manufacturing: The replacement of expensive, specialized chiral resolving agents with bulk amino acids results in a drastic reduction in raw material costs. Additionally, the high selectivity of the new resolution method minimizes the loss of valuable starting material to the mother liquor, improving overall mass balance and yield. The elimination of extensive purification steps required to remove stubborn stereoisomers further reduces solvent consumption and waste disposal costs, contributing to a leaner and more economically viable production model.
- Enhanced Supply Chain Reliability: By utilizing resolving agents that are classified as nutritional supplements or food additives, the manufacturing process gains access to a robust and diversified global supply network. This reduces the risk of production stoppages caused by shortages of niche fine chemicals. The stability of these raw materials also simplifies storage and handling requirements, ensuring consistent quality input for the synthesis of high-purity antineoplastic intermediates regardless of geopolitical or logistical fluctuations.
- Scalability and Environmental Compliance: The mild reaction conditions employed in this protocol, particularly the avoidance of harsh bases or extreme temperatures during resolution, facilitate seamless commercial scale-up of complex peptide intermediates. The process generates less hazardous waste compared to traditional methods involving heavy metal catalysts or toxic solvents, aligning with modern green chemistry principles. This environmental compatibility simplifies regulatory compliance and permits faster approval for expanded production capacities, ensuring reducing lead time for high-purity pharmaceutical intermediates to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical advantages of adopting this methodology for large-scale production.
Q: Why are L-lysine and L-arginine preferred over S-(-)-Alpha-Methyl benzylamine for resolution?
A: Traditional resolving agents like S-(-)-Alpha-Methyl benzylamine often fail to completely separate the (2S,3R) and (2R,3S) enantiomers, leading to persistent optical impurities. The use of basic amino acids like L-lysine allows for more effective salification and crystallization, achieving enantiomeric excess (ee) values exceeding 99.5%.
Q: How does the EDCI/HOAt system improve product quality compared to DCC/HOBt?
A: Conventional DCC/HOBt coupling can induce racemization at the C-5 chiral center during peptide bond formation, generating inactive isomers. The EDCI/HOAt combination significantly accelerates the reaction rate while strictly maintaining the stereochemical integrity of the leucine moiety, minimizing trace impurities.
Q: What is the expected purity profile of Ubenimex produced via this method?
A: By combining high-efficiency chiral resolution with racemization-suppressed coupling, this process yields Ubenimex with a chemical purity greater than 99.5%, effectively eliminating the pharmacologically inactive stereoisomers that compromise therapeutic efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ubenimex Supplier
As the pharmaceutical landscape evolves towards stricter purity standards and more sustainable manufacturing practices, partnering with a technically adept CDMO is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate chiral resolution and coupling steps described in Patent CN101891647A are executed with precision. Our facility is equipped with rigorous QC labs capable of detecting trace stereoisomers, guaranteeing that every batch meets stringent purity specifications required for oncology applications. We understand that the consistency of your final drug product depends entirely on the quality of the intermediates we supply.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this amino acid-mediated process. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver reliable, high-quality Ubenimex intermediates that support your clinical and commercial goals.
