Advanced Organocatalytic Kinetic Resolution for High-Value Chiral Tertiary Alcohols and Dihydroisobenzofurans
Advanced Organocatalytic Kinetic Resolution for High-Value Chiral Tertiary Alcohols and Dihydroisobenzofurans
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access complex chiral scaffolds, particularly those containing sterically hindered tertiary alcohol motifs which are prevalent in numerous bioactive molecules. Patent CN113979975A introduces a groundbreaking kinetic resolution method that utilizes a specialized chiral phosphoric acid catalyst to transform racemic aryl allyl tertiary alcohols into high-value enantiomerically enriched products. This technology represents a significant paradigm shift away from traditional transition-metal-catalyzed asymmetric additions, offering a metal-free alternative that aligns perfectly with the stringent purity requirements of modern active pharmaceutical ingredient (API) manufacturing. By leveraging the unique steric and electronic properties of bulky binaphthol-derived phosphoric acids, this process achieves exceptional stereocontrol while simultaneously generating two distinct classes of valuable intermediates: chiral tertiary alcohols and polysubstituted dihydroisobenzofurans.
The core innovation lies in the ability of the chiral phosphoric acid to differentiate between the enantiomers of the sterically crowded tertiary alcohol substrate through a highly organized hydrogen-bonding network. Unlike previous methods that struggled with the low reactivity of tertiary alcohols due to steric hindrance, this novel catalytic system effectively activates the hydroxyl group, facilitating a dynamic kinetic resolution pathway. The reaction proceeds under remarkably mild conditions, typically utilizing chloroform as a solvent and molecular sieves as an essential additive to sequester water, which drives the equilibrium forward. This approach not only solves the long-standing challenge of resolving tertiary alcohols without enzymes but also provides a versatile platform for synthesizing diverse chiral building blocks that are critical for the development of next-generation therapeutics and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral diaryl tertiary alcohol structures has relied heavily on transition metal-catalyzed asymmetric addition reactions, such as the addition of organoboron reagents to ketones. While these methods can be effective, they introduce significant downstream processing challenges, particularly regarding the removal of toxic metal residues like rhodium or palladium to parts-per-million levels required by regulatory bodies. Furthermore, these transition metal processes often suffer from limited functional group tolerance, necessitating extensive protecting group strategies that increase step count and reduce overall atom economy. The reliance on expensive noble metals also creates supply chain vulnerabilities and cost volatility, making the final API intermediates significantly more expensive to produce. Additionally, traditional kinetic resolution methods for tertiary alcohols have been scarce and often inefficient, as the steric bulk around the reaction center typically hinders the approach of the catalyst, leading to poor conversion rates and low enantioselectivity.
The Novel Approach
In stark contrast, the method disclosed in CN113979975A employs a sophisticated chiral phosphoric acid catalyst that operates entirely without transition metals, thereby eliminating the need for costly metal scavenging units and simplifying the purification workflow. This organocatalytic strategy utilizes a specifically designed binaphthol backbone substituted with bulky groups, such as tert-butyldiphenylsilyl (TBDPS) and tricyclohexylphenyl moieties, to create a rigid chiral pocket that effectively discriminates between substrate enantiomers. The reaction conditions are exceptionally mild, often proceeding at temperatures as low as 0°C, which minimizes thermal degradation of sensitive functional groups and reduces energy consumption. Moreover, this single-step transformation concurrently produces two valuable chiral outputs—the unreacted tertiary alcohol and the cyclized dihydroisobenzofuran—effectively doubling the utility of the starting material and enhancing the overall process economics compared to linear synthetic routes that discard half of the racemic mixture.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Kinetic Resolution
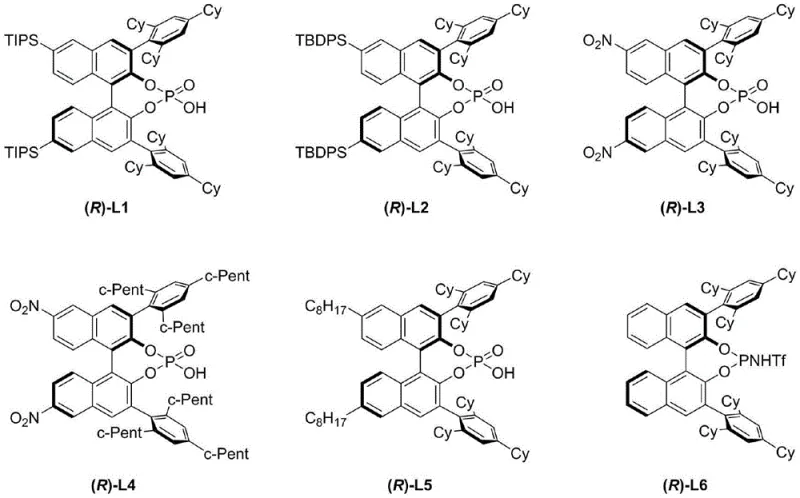
The mechanistic success of this transformation hinges on the dual activation capability of the chiral phosphoric acid, which acts as a Brønsted acid to protonate the hydroxyl group of the tertiary alcohol while simultaneously engaging in hydrogen bonding with the leaving group or the developing carbocation intermediate. The bulky substituents at the 3,3'-positions of the binaphthyl skeleton, such as the tricyclohexylphenyl groups found in catalyst (R)-L1, create a confined chiral environment that imposes severe steric constraints on the approaching nucleophile or the departing water molecule. This steric differentiation is crucial for distinguishing the pro-chiral faces of the planar carbocation-like intermediate formed during the ionization of the tertiary alcohol. The catalyst essentially acts as a chiral shield, allowing only one enantiomer of the substrate to react rapidly via an SN1-type pathway to form the dihydroisobenzofuran ring, while the other enantiomer remains largely unreacted, thus achieving kinetic resolution.
Furthermore, the presence of molecular sieves plays a pivotal role in the reaction mechanism by irreversibly adsorbing the water molecule generated during the cyclization process. This removal of water prevents the reverse hydrolysis reaction, effectively driving the equilibrium towards the formation of the dihydroisobenzofuran product and ensuring high conversion rates despite the reversible nature of the initial ionization step. The electronic effects of the substituents on the catalyst backbone also fine-tune the acidity of the phosphoric acid, optimizing the balance between substrate activation and catalyst stability. For instance, the introduction of TBDPS groups at the 6,6'-positions enhances the rigidity of the catalyst framework, preventing conformational flexibility that could otherwise erode enantioselectivity. This precise tuning of steric and electronic parameters allows the system to overcome the inherent difficulty of differentiating three non-hydrogen substituents at a quaternary stereocenter, a feat that has historically plagued asymmetric synthesis efforts.
How to Synthesize Chiral Aryl Allyl Tertiary Alcohols Efficiently
The practical implementation of this kinetic resolution protocol involves a straightforward setup that is amenable to standard laboratory and pilot plant equipment. The process begins with the rigorous drying of the reaction vessel and the activation of molecular sieves to ensure an anhydrous environment, which is critical for maintaining the activity of the chiral phosphoric acid catalyst. The substrate, a racemic aryl allyl tertiary alcohol, is dissolved in dry chloroform, and the catalyst is added at a loading of approximately 15 mol%, a concentration that balances cost efficiency with reaction rate. The reaction is typically initiated at 0°C and monitored closely via high-performance liquid chromatography (HPLC) to determine the optimal stopping point for maximum enantiomeric enrichment. Detailed standardized synthesis steps for the preparation of the catalyst and the execution of the resolution reaction are provided in the guide below.
- Prepare the reaction system under nitrogen protection using anhydrous chloroform as the solvent and activate 4Å molecular sieves as the additive.
- Add the racemic aryl allyl tertiary alcohol substrate and the chiral binaphthol-derived phosphoric acid catalyst (e.g., (R)-L1) at 0°C.
- Monitor the reaction progress via HPLC until completion, then filter through celite and purify the resulting chiral tertiary alcohol and dihydroisobenzofuran products via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic kinetic resolution technology offers profound strategic advantages that extend far beyond simple yield metrics. By transitioning from transition-metal-dependent processes to this metal-free organocatalytic system, manufacturers can drastically simplify their supply chain logistics by removing the dependency on volatile noble metal markets and the complex regulatory documentation associated with heavy metal usage. The elimination of metal catalysts means that the costly and time-consuming steps of metal scavenging, filtration through specialized resins, and extensive ICP-MS testing are no longer required, leading to a streamlined production workflow that significantly reduces the overall manufacturing lead time. This simplification directly translates to enhanced supply chain reliability, as the risk of batch failure due to metal contamination is virtually eliminated, ensuring a consistent and uninterrupted flow of high-purity intermediates to downstream formulation teams.
- Cost Reduction in Manufacturing: The economic impact of switching to this chiral phosphoric acid catalyzed process is substantial, primarily driven by the removal of expensive transition metal catalysts and the associated purification infrastructure. Traditional methods often require stoichiometric or near-stoichiometric amounts of precious metals like rhodium or palladium, along with specialized ligands that are costly to synthesize and sensitive to air and moisture. In contrast, the organocatalyst used in this patent is robust, air-stable, and can be synthesized from readily available binaphthol precursors, leading to a drastic reduction in raw material costs. Furthermore, the ability to isolate two valuable chiral products from a single reaction pot effectively halves the cost burden per unit of chiral material produced, as the 'waste' product in traditional kinetic resolutions is here converted into a high-value dihydroisobenzofuran scaffold that can be sold or utilized in parallel synthesis streams.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this chemical process ensures greater continuity of supply, which is critical for meeting the just-in-time delivery demands of global pharmaceutical clients. The reagents involved, such as chloroform and molecular sieves, are commodity chemicals with stable global availability, unlike specialized organometallic complexes that may face supply bottlenecks. The mild reaction conditions, operating near room temperature or slightly below, reduce the energy load on production facilities and minimize the wear and tear on reactor vessels, thereby extending equipment lifespan and reducing maintenance downtime. This operational stability allows for more predictable production scheduling and reduces the risk of unexpected shutdowns, providing procurement teams with the confidence to commit to long-term supply agreements with key customers.
- Scalability and Environmental Compliance: The environmental profile of this synthesis route is exceptionally favorable, aligning with the increasing pressure on chemical manufacturers to adopt greener technologies and reduce their carbon footprint. The reaction generates water as the only stoichiometric byproduct in the cyclization pathway, resulting in high atom economy and minimizing the volume of hazardous waste that requires treatment and disposal. The absence of heavy metals simplifies wastewater treatment protocols and reduces the regulatory burden associated with discharging metal-containing effluents. Moreover, the process is inherently scalable; the use of heterogeneous additives like molecular sieves facilitates easy separation via filtration, a unit operation that scales linearly from gram to ton quantities without the mixing and heat transfer issues often encountered with homogeneous transition metal catalysis. This scalability ensures that the technology can seamlessly transition from R&D optimization to commercial scale-up of complex pharmaceutical intermediates without requiring fundamental process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral phosphoric acid catalyzed kinetic resolution technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable foundation for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing synthetic portfolios and for procurement specialists assessing the total cost of ownership. We encourage stakeholders to review these insights to fully appreciate the transformative potential of this metal-free methodology in the context of modern fine chemical manufacturing.
Q: What are the primary advantages of using chiral phosphoric acid over transition metal catalysts for tertiary alcohol resolution?
A: The primary advantage is the complete elimination of transition metals, which removes the need for expensive and complex heavy metal scavenging steps required to meet strict pharmaceutical purity standards. Additionally, this organocatalytic method operates under milder conditions with higher functional group tolerance.
Q: What is the typical enantiomeric excess (ee) achievable with this kinetic resolution method?
A: The method demonstrates excellent stereoselectivity, consistently achieving enantiomeric excess (ee) values ranging from 85% to 97% for the recovered tertiary alcohol products and up to 95% for the cyclized dihydroisobenzofuran byproducts, depending on the specific substrate substituents.
Q: Can this process be scaled for commercial production of API intermediates?
A: Yes, the process is highly scalable due to its use of stable, non-toxic organic solvents like chloroform and robust solid additives like molecular sieves. The absence of air-sensitive transition metal complexes simplifies the engineering requirements for large-scale reactor operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tertiary Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral phosphoric acid catalyzed kinetic resolution technology described in CN113979975A for producing high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise determination of enantiomeric excess (ee) values using advanced chiral HPLC methods, guaranteeing that every batch meets the exacting standards required by global regulatory agencies. We are committed to leveraging this innovative organocatalytic chemistry to deliver cost-effective and sustainable solutions for our partners.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how switching to this metal-free process can improve your bottom line. Please contact us to request specific COA data for similar chiral tertiary alcohol intermediates and to discuss route feasibility assessments for your target molecules. Let us help you engineer a more resilient and profitable supply chain for your critical drug substances.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
