Advanced Copper-Catalyzed Synthesis of Polysubstituted 2-Aryl Indole Derivatives for Pharmaceutical Applications
Advanced Copper-Catalyzed Synthesis of Polysubstituted 2-Aryl Indole Derivatives for Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A significant breakthrough in this domain is detailed in patent CN111004164A, which discloses a robust preparation method for polysubstituted 2-aryl indole derivatives. This technology addresses critical bottlenecks in the synthesis of indole-based scaffolds, which are ubiquitous in medicinal chemistry due to their profound biological activities. The indole nucleus serves as a privileged structure in drug discovery, acting as a key pharmacophore in numerous therapeutic agents ranging from anti-inflammatories to anticancer drugs. For instance, Zindoxifene, a selective estrogen receptor modulator, relies heavily on the 2-aryl indole framework for its efficacy.
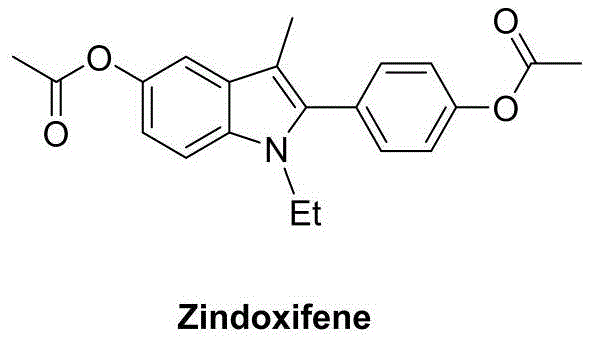
As illustrated by the structure of Zindoxifene, the precise substitution pattern on the indole ring is crucial for biological activity. Traditional methods often struggle to introduce these substituents with high regioselectivity and yield without resorting to expensive catalysts or hazardous reagents. The methodology outlined in CN111004164A offers a transformative solution by utilizing a copper-catalyzed oxidative cyclization strategy. This approach not only streamlines the synthetic pathway but also enhances the economic viability of producing high-value pharmaceutical intermediates. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this catalytic system is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-aryl indole skeleton has been fraught with challenges that hinder large-scale commercial production. Early methodologies often relied on the use of noble metal catalysts such as palladium or iridium, which are not only prohibitively expensive but also subject to volatile market pricing and supply chain disruptions. For example, prior art describes methods involving Fe and Pd synergistic catalysis for intramolecular hydroamination, followed by Michael addition. While chemically feasible, these routes suffer from low yields and the necessity of using toxic reagents like methyl vinyl ketone, posing significant safety and environmental hazards in an industrial setting. Furthermore, iridium-catalyzed approaches often require specialized, difficult-to-synthesize ligands, adding layers of complexity and cost to the process. These limitations result in extended lead times and inflated production costs, making it difficult for manufacturers to compete in the global market for high-purity OLED material or pharmaceutical precursors.
The Novel Approach
In stark contrast, the novel approach presented in the patent leverages abundant and inexpensive copper salts to drive the oxidative cyclization reaction. By employing simple, commercially available ligands such as 2'-bipyridine or 1,10-phenanthroline, the method eliminates the dependency on scarce noble metals. The reaction proceeds through a direct coupling of 2-alkynylanilines and ketones in the presence of an oxidant like TEMPO or 4-OH-TEMPO. This one-pot transformation is operationally simple, requiring standard heating conditions (typically 120 °C) in common solvents like 1,2-dichlorobenzene. The result is a highly efficient process that delivers polysubstituted 2-aryl indoles with excellent chemical and regioselectivity. This shift from precious metal catalysis to base metal catalysis represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, offering a sustainable pathway for the commercial scale-up of complex heterocycles.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the intricate interplay between the copper catalyst, the nitrogen-containing ligand, and the nitroxyl radical oxidant. The reaction initiates with the coordination of the copper species to the alkyne moiety of the 2-alkynylaniline substrate, activating it towards nucleophilic attack. Simultaneously, the amine group participates in the cyclization process, facilitated by the oxidative environment created by the TEMPO derivative. The oxidant plays a dual role: it regenerates the active copper species to sustain the catalytic cycle and facilitates the aromatization of the intermediate dihydroindole to the final indole product. This mechanism ensures that the reaction proceeds with high atom economy and minimal byproduct formation. The use of 4-OH-TEMPO, in particular, has been shown to enhance reaction efficiency, likely due to its specific redox potential and solubility characteristics in the reaction medium.

From an impurity control perspective, this mechanism offers distinct advantages. The high regioselectivity of the copper-catalyzed cyclization minimizes the formation of structural isomers, which are often difficult to separate and can compromise the purity profile of the final API. The reaction conditions are sufficiently mild to prevent the decomposition of sensitive functional groups, such as halogens or methoxy groups, which are frequently present in drug candidates. As shown in the general reaction scheme, the versatility of the method allows for the incorporation of diverse R1, R2, and R3 groups, enabling the synthesis of a broad array of derivatives. This flexibility is paramount for medicinal chemists who need to rapidly generate analog libraries for structure-activity relationship (SAR) studies. The robustness of the catalytic system ensures consistent quality across different batches, a critical factor for maintaining stringent purity specifications in regulated industries.
How to Synthesize Polysubstituted 2-Aryl Indole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The protocol involves dissolving the 2-alkynyl aniline and ketone substrates in a high-boiling solvent, followed by the addition of the copper catalyst and ligand system. The mixture is then heated under an oxidative atmosphere provided by the TEMPO reagent. Optimization of the molar ratios is key; typically, a slight excess of the ketone (1:1.2 ratio) drives the reaction to completion. Post-reaction workup is straightforward, involving solvent removal and purification via standard column chromatography.
- Charge a reactor with 2-alkynyl aniline derivative, ketone compound, copper catalyst (e.g., Cu(OAc)2), ligand (e.g., 2'-bipyridine), and 4-OH-TEMPO oxidant in a solvent like 1,2-dichlorobenzene.
- Heat the reaction mixture to a temperature between 80-120 °C, preferably 120 °C, and maintain stirring for 24 to 36 hours to ensure complete oxidative cyclization.
- Upon completion, recover the solvent by concentration under reduced pressure and purify the crude product via column chromatography using n-hexane/ethyl acetate to isolate the target indole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary driver is the drastic simplification of the raw material portfolio. By replacing expensive palladium or iridium catalysts with commodity copper salts, companies can achieve substantial cost savings in their bill of materials. Additionally, the ligands required, such as bipyridines, are bulk chemicals with stable supply chains, reducing the risk of production stoppages due to material shortages. The elimination of toxic reagents like methyl vinyl ketone also lowers the barrier for regulatory compliance and waste disposal, further contributing to overall cost reduction in API manufacturing. These factors combine to create a more resilient and economical supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with inexpensive copper salts fundamentally alters the cost structure of the synthesis. Copper is orders of magnitude cheaper than palladium or iridium, and the ligand systems used are simple, off-the-shelf chemicals. This shift eliminates the need for costly metal scavenging steps often required to meet residual metal limits in pharmaceutical products. Consequently, the overall processing costs are significantly reduced, allowing for more competitive pricing of the final intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials such as substituted anilines and ketones ensures a stable supply chain. Unlike specialized organometallic reagents that may have long lead times or single-source suppliers, the inputs for this reaction are produced globally in large volumes. This abundance mitigates the risk of supply disruptions and allows for flexible sourcing strategies. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, enhancing the reliability of production schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The operational simplicity of the method makes it highly amenable to scale-up. The reaction uses standard solvents and temperatures that are easily managed in existing reactor infrastructure, avoiding the need for specialized high-pressure or cryogenic equipment. From an environmental standpoint, the use of TEMPO oxidants generates benign byproducts compared to stoichiometric heavy metal oxidants. This aligns with green chemistry principles, simplifying waste treatment processes and reducing the environmental footprint of the manufacturing facility, which is increasingly important for meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear picture of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product pipelines.
Q: Why is copper catalysis preferred over palladium for indole synthesis?
A: Copper catalysts are significantly more cost-effective than noble metals like palladium or iridium. The patented method utilizes inexpensive copper salts (e.g., copper acetate) combined with simple ligands, drastically reducing raw material costs while maintaining high chemical selectivity and regioselectivity compared to traditional precious metal methods.
Q: What represents the key advantage of this oxidative cyclization method?
A: The primary advantage lies in the direct one-pot construction of the indole skeleton from readily available 2-alkynylanilines and ketones. Unlike older methods requiring toxic reagents like methyl vinyl ketone or complex multi-step sequences, this approach uses mild oxidants like TEMPO, simplifying the workflow and improving overall process safety and efficiency.
Q: Can this method accommodate diverse substrate substitutions?
A: Yes, the method demonstrates excellent substrate tolerance. It successfully accommodates various substituents on both the aniline and ketone components, including electron-donating groups like methoxy and electron-withdrawing groups like fluoro, chloro, and bromo, allowing for the synthesis of a wide library of polysubstituted 2-aryl indole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 2-Aryl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. Our team of expert chemists has thoroughly analyzed the copper-catalyzed oxidative cyclization route described in CN111004164A and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of polysubstituted 2-aryl indole meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By partnering with us, you gain access to our deep technical expertise and state-of-the-art production capabilities. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can deliver value to your organization, ensuring a reliable supply of high-quality pharmaceutical intermediates for your global operations.
