Advanced Metal-Free Synthesis of Quinoline-4-Carboxylates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and compliant pathways for synthesizing critical heterocyclic scaffolds. Patent CN109336811B introduces a groundbreaking preparation method for quinoline-4-carboxylate compounds, a class of molecules pivotal in the development of antimalarial agents and other therapeutic drugs. This technology leverages a bulky borane catalyst to facilitate a one-pot multi-component reaction, effectively merging aniline derivatives, aldehydes, and pyruvate esters into the desired quinoline structure. Unlike traditional methods that rely heavily on transition metals, this innovation utilizes a non-metallic catalytic system, addressing the growing regulatory pressure regarding heavy metal residues in active pharmaceutical ingredients. The significance of this patent lies not only in its chemical elegance but also in its potential to streamline the supply chain for high-purity pharmaceutical intermediates, offering a robust solution for manufacturers aiming to enhance both product quality and process sustainability.
Quinoline-4-carboxylates serve as essential building blocks in medicinal chemistry, notably as precursors for dihydroorotate dehydrogenase inhibitors which have shown promise in treating malaria and other diseases. The conventional synthesis of these compounds has often been plagued by limitations such as harsh reaction conditions, narrow substrate scope, and the inevitable introduction of metal contaminants. The technology disclosed in CN109336811B overcomes these hurdles by employing a Lewis acidic borane catalyst that operates under mild conditions, typically between 0°C and 100°C. This approach not only improves the overall yield, which can reach up to 95% in specific embodiments, but also ensures that the final product is free from toxic metal traces, thereby reducing the burden on downstream purification processes and ensuring compliance with stringent international pharmacopoeia standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline-4-carboxylate derivatives has relied on various transition metal catalysts, each presenting distinct challenges for large-scale manufacturing. For instance, earlier methodologies utilized ytterbium trifluoromethanesulfonate (Yb(OTf)3) to catalyze the reaction between aldehyde derivatives and naphthylamines. While this method demonstrated good activity towards aromatic aldehydes with yields ranging from 76% to 92%, it suffered from a significant inability to process aliphatic aldehydes, thereby restricting its utility in diverse chemical libraries. Furthermore, the use of rare earth metals like ytterbium introduces concerns regarding cost volatility and the environmental impact of mining and processing these elements. The presence of such metals in the final reaction mixture necessitates rigorous and often expensive purification steps to meet the strict ppm limits imposed by regulatory bodies for pharmaceutical products.
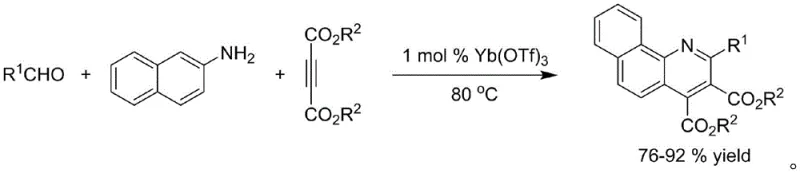
Other prior art methods have explored visible-light-induced radical reactions using iridium catalysts or cooperative catalysis systems involving zirconium and copper. Although these approaches offered improvements in reaction conditions, such as room temperature operation, they still fundamentally depended on precious or heavy metals. The iridium-catalyzed routes, for example, often resulted in moderate yields between 40% and 77% and required specialized equipment for light irradiation, adding complexity to the reactor setup. Similarly, zirconium and copper cooperative systems, while effective, introduced multiple metal species into the reaction pot, complicating the waste treatment process and increasing the risk of cross-contamination. These conventional methods collectively highlight a critical industry pain point: the trade-off between reaction efficiency and the operational burden of metal removal, which directly impacts the cost of goods sold and the speed to market for new drug candidates.
The Novel Approach
In stark contrast to the metal-dependent legacy processes, the novel approach detailed in patent CN109336811B utilizes a large sterically hindered borane catalyst, such as tris(pentafluorophenyl)borane, to drive the multi-component coupling. This metal-free strategy represents a paradigm shift in heterocyclic synthesis, enabling the efficient construction of the quinoline core without the introduction of any transition metal species. The reaction proceeds smoothly in common organic solvents like chloroform or toluene, demonstrating exceptional tolerance to both moisture and air, which significantly simplifies the operational requirements for plant operators. By eliminating the need for inert atmosphere gloveboxes or rigorous solvent drying, this method reduces the capital expenditure associated with specialized reactor infrastructure and lowers the energy consumption typically required for maintaining strict anhydrous conditions.
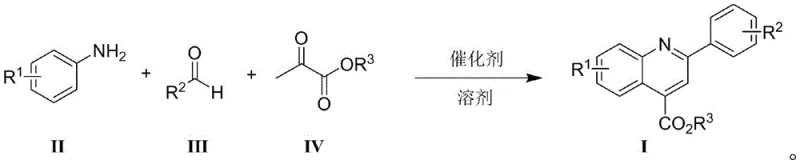
The versatility of this new catalytic system is evidenced by its broad substrate scope, successfully accommodating a wide array of aniline compounds bearing different functional groups such as fluoro, chloro, trifluoromethyl, and methoxy substituents. Furthermore, it effectively processes both aromatic and aliphatic aldehydes, overcoming the limitations seen in earlier ytterbium-catalyzed methods. The one-pot nature of the reaction ensures high atomic economy, as all three components—aniline, aldehyde, and pyruvate—are combined in a single vessel to directly yield the target quinoline-4-carboxylate. This consolidation of steps not only minimizes solvent usage and waste generation but also drastically reduces the labor hours required for intermediate isolation and handling, presenting a compelling value proposition for procurement teams focused on optimizing manufacturing efficiency and reducing the environmental footprint of chemical production.
Mechanistic Insights into Borane-Catalyzed Multi-Component Cyclization
The efficacy of the borane-catalyzed synthesis lies in the unique Lewis acidic properties of the bulky boron species, which activate the carbonyl and imine intermediates without forming stable coordination complexes that could lead to catalyst deactivation. In the proposed mechanism, the borane catalyst initially coordinates with the oxygen atom of the aldehyde or the pyruvate ester, increasing the electrophilicity of the carbonyl carbon. This activation facilitates the nucleophilic attack by the aniline derivative, leading to the formation of an imine intermediate in situ. Subsequently, the activated pyruvate ester undergoes a Mannich-type addition to the imine, followed by a cyclization and oxidation sequence that aromatizes the ring system to form the stable quinoline structure. The steric bulk of the catalyst, particularly in the case of tris(pentafluorophenyl)borane, prevents the formation of unreactive adducts, ensuring that the catalytic cycle turns over rapidly even at low catalyst loadings of 0.1 mol%.
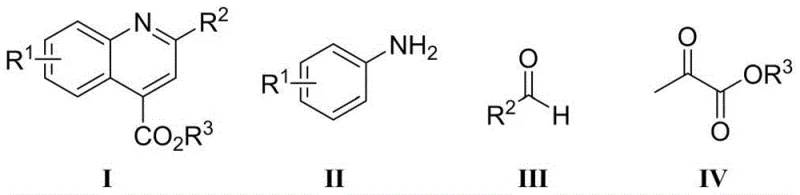
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed pathways. Transition metal catalysts often promote side reactions such as homocoupling or over-oxidation, leading to complex impurity profiles that are difficult to separate. The borane-catalyzed route, operating under milder thermal conditions (preferably 30°C to 60°C), minimizes thermal degradation of sensitive functional groups and suppresses the formation of polymeric byproducts. The high selectivity of the reaction ensures that the crude product contains fewer impurities, simplifying the downstream purification process which typically involves standard silica gel column chromatography. For R&D directors, this implies a more predictable scale-up trajectory, as the reaction kinetics are less sensitive to minor fluctuations in temperature or mixing efficiency, thereby ensuring consistent batch-to-batch quality and reducing the risk of failed production runs due to out-of-specification impurity levels.
How to Synthesize Quinoline-4-Carboxylates Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and purity. The process begins by dissolving the aniline derivative, aldehyde, and pyruvate ester in a suitable organic solvent, with chloroform being the preferred medium due to its solubility profile and boiling point. The bulky borane catalyst is then introduced to the mixture, and the reaction is allowed to proceed with stirring. Detailed standard operating procedures regarding specific molar ratios, temperature ramps, and work-up protocols are essential for reproducibility. For a comprehensive guide on the exact experimental conditions and troubleshooting tips for specific substrates, please refer to the standardized synthesis steps provided below.
- Dissolve aniline derivatives, aldehyde compounds, and pyruvate esters in an organic solvent such as chloroform or toluene.
- Add a bulky borane catalyst, preferably tris(pentafluorophenyl)borane, to the reaction mixture under ambient atmosphere.
- Stir the reaction at mild temperatures between 30°C and 60°C for 2 to 12 hours, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology translates into tangible operational benefits that extend beyond simple yield improvements. The primary advantage is the drastic simplification of the purification workflow. In traditional metal-catalyzed processes, a significant portion of the production budget is allocated to scavenging resins, filtration units, and analytical testing required to verify that heavy metal levels are below regulatory thresholds. By completely eliminating the use of transition metals from the outset, this method removes the need for these costly and time-consuming remediation steps. This results in a streamlined production cycle where the time from reaction completion to final product release is significantly shortened, allowing for faster inventory turnover and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of switching to a borane-catalyzed system is profound, driven primarily by the reduction in raw material costs and waste disposal fees. Transition metal catalysts, especially those based on iridium or palladium, are subject to significant price volatility and supply constraints. Replacing these with organoboron compounds, which are generally more stable and cost-effective, stabilizes the bill of materials. Additionally, the absence of metal residues means that solvent recovery streams are cleaner and can be recycled more easily, further reducing the consumption of fresh solvents. The high atom economy of the one-pot reaction also ensures that raw materials are converted into product with minimal waste, leading to substantial cost savings in raw material procurement and waste treatment operations.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the catalytic system against environmental factors. The tolerance of the borane catalyst to moisture and air means that the reaction does not require strictly anhydrous conditions or inert gas blanketing, which are often points of failure in large-scale reactors. This robustness reduces the risk of batch failures due to equipment leaks or operator error, ensuring a more consistent supply of the intermediate. Furthermore, the reagents involved—anilines, aldehydes, and pyruvates—are commodity chemicals with well-established global supply chains, reducing the risk of bottlenecks associated with sourcing specialized or proprietary catalysts. This availability ensures that production schedules can be maintained without interruption, even in times of global supply chain stress.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the mild reaction conditions and the exothermic profile of the reaction, which is manageable with standard cooling systems. The elimination of heavy metals aligns perfectly with increasingly stringent environmental regulations regarding effluent discharge and soil contamination. Facilities adopting this technology can significantly reduce their environmental compliance burden, avoiding the costs associated with treating heavy metal-containing wastewater. The green chemistry metrics of the process, including high yield and low E-factor, make it an attractive option for companies aiming to improve their sustainability scores and meet corporate social responsibility goals, thereby enhancing the brand value of the final pharmaceutical products derived from these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this borane-catalyzed synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for stakeholders evaluating the potential integration of this method into their existing manufacturing portfolios.
Q: Why is the borane catalyst preferred over traditional metal catalysts for quinoline synthesis?
A: The borane catalyst eliminates the risk of heavy metal contamination, which is critical for pharmaceutical intermediates. It also offers superior tolerance to moisture and air, simplifying operational requirements compared to sensitive metal complexes.
Q: What is the substrate scope of this new preparation method?
A: The method demonstrates broad applicability, successfully accommodating aromatic amines with various electron-donating or withdrawing groups, as well as both aromatic and aliphatic aldehydes, yielding up to 95% in optimized conditions.
Q: How does this method impact the cost of manufacturing quinoline-4-carboxylates?
A: By removing the need for expensive transition metals and the subsequent purification steps required to remove metal residues, the overall production cost is significantly reduced while maintaining high atomic economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline-4-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our technical team has thoroughly evaluated the borane-catalyzed route described in CN109336811B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of quinoline-4-carboxylate we supply meets the highest standards required for drug substance synthesis. We are committed to delivering high-purity pharmaceutical intermediates that enable our partners to accelerate their drug development timelines with confidence.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains using this cutting-edge technology. By leveraging our expertise in process chemistry, we can help you implement this metal-free route to achieve significant efficiency gains. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us partner to engineer a more sustainable and cost-effective future for your chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
