Scalable Metal-Free Synthesis of Quinoline-4-Carboxylates for Global Pharma Supply Chains
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, safer synthetic methodologies, particularly for critical heterocyclic intermediates like quinoline-4-carboxylates. Patent CN109336811B discloses a groundbreaking preparation method that utilizes large sterically hindered borane catalysts to facilitate a multi-component one-pot reaction. This innovation addresses the long-standing challenge of heavy metal contamination inherent in traditional transition metal-catalyzed processes. By replacing toxic metals with non-metallic borane species, this technology not only enhances the purity profile of the resulting intermediates but also streamlines the downstream purification workflow. For global supply chain leaders, this represents a significant opportunity to secure a more robust and compliant source of antimalarial and kinase inhibitor precursors. The method's ability to operate under mild conditions while maintaining high atom economy positions it as a superior alternative for commercial-scale API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline-4-carboxylate derivatives has relied heavily on transition metal catalysts, which introduce substantial regulatory and operational burdens. For instance, earlier methodologies employed Ytterbium trifluoromethanesulfonate (Yb(OTf)3) to catalyze the reaction between aldehyde derivatives and naphthylamines, as illustrated in the reaction scheme below. While these methods achieved moderate yields, they were fundamentally limited by their inability to process aliphatic aldehydes effectively and, more critically, by the risk of heavy metal leaching.
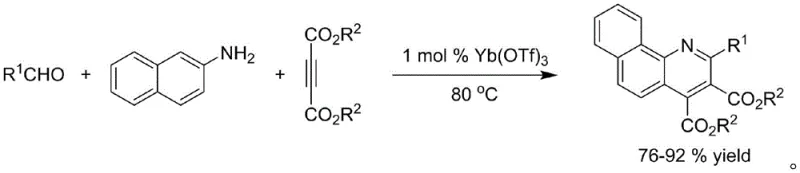
Furthermore, other prior art techniques utilized visible-light promoted Iridium catalysis or cooperative Zirconocene/Copper systems. These approaches often require stringent anhydrous conditions, specialized equipment for light irradiation, or complex multi-step workups to remove residual metals. The presence of heavy metals like Iridium, Copper, or Ytterbium necessitates additional purification stages, such as chromatography or specialized scavenging resins, which drastically increase production costs and extend lead times. For procurement managers, these hidden costs and the risk of batch rejection due to metal specifications make conventional routes less attractive for long-term supply contracts.
The Novel Approach
In stark contrast, the novel approach detailed in CN109336811B employs a large sterically hindered borane catalyst to drive the multi-component condensation of anilines, aldehydes, and pyruvates. This metal-free strategy fundamentally alters the reaction landscape by eliminating the source of heavy metal contamination at the very beginning of the synthesis. The general reaction scheme demonstrates how the three components converge in a single pot to form the target quinoline-4-carboxylate structure with high efficiency.
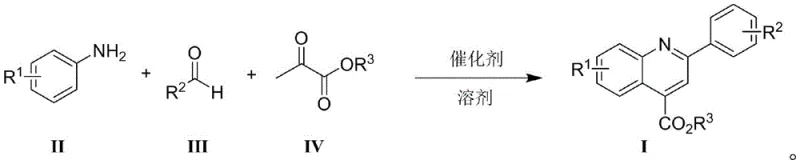
This methodology offers a distinct advantage in terms of substrate tolerance and operational simplicity. Unlike previous metal-catalyzed systems that failed with aliphatic aldehydes or required specific electronic properties on the aromatic rings, the borane-catalyzed system exhibits broad applicability. It successfully accommodates various functional groups including fluoro, chloro, and trifluoromethyl substituents without compromising yield. This flexibility allows manufacturers to produce a diverse library of analogues using a unified platform, significantly reducing the need for process re-optimization when scaling up new derivatives for clinical trials.
Mechanistic Insights into Borane-Catalyzed Cyclization
The core of this technological breakthrough lies in the unique Lewis acidity of the bulky borane catalyst, specifically tris(pentafluorophenyl)borane (B(C6F5)3). This catalyst activates the carbonyl group of the aldehyde and the pyruvate ester through coordination, facilitating the nucleophilic attack by the aniline without the need for transition metal redox cycles. The specific reaction conditions utilizing B(C6F5)3 in chloroform at moderate temperatures highlight the precision of this activation mechanism.
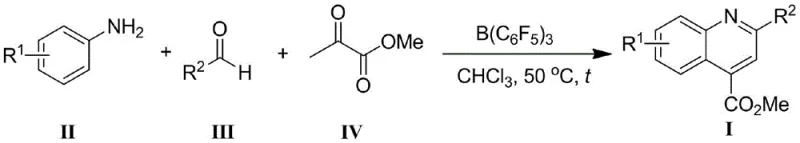
From an impurity control perspective, the absence of transition metals simplifies the impurity profile significantly. Traditional metal-catalyzed reactions often generate metal-organic byproducts or require ligands that can persist through purification. In this metal-free system, the primary impurities are unreacted starting materials or simple condensation byproducts, which are far easier to remove via standard crystallization or washing techniques. This results in a final product with superior purity specifications, directly addressing the concerns of R&D directors regarding the quality and consistency of critical starting materials for drug synthesis.
How to Synthesize Quinoline-4-Carboxylate Efficiently
To implement this synthesis effectively, the process involves dissolving the aniline, aldehyde, and pyruvate components in a suitable organic solvent such as chloroform or toluene. The key operational parameter is the addition of the bulky borane catalyst at a molar ratio as low as 0.1 mol%, which is sufficient to drive the reaction to completion. The mixture is then stirred at temperatures ranging from 30°C to 60°C for a period of 2 to 12 hours, depending on the specific substrate reactivity.
- Dissolve aniline derivatives, aldehyde compounds, and pyruvate esters in an organic solvent such as chloroform or toluene.
- Add a large sterically hindered borane catalyst, preferably tris(pentafluorophenyl)borane, to the reaction mixture.
- Stir the reaction at mild temperatures between 30°C and 60°C for 2 to 12 hours to achieve high conversion yields.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free protocol offers tangible strategic benefits beyond mere chemical efficiency. The elimination of expensive transition metal catalysts such as Iridium or Ytterbium directly reduces the raw material cost base. Moreover, the removal of metal scavenging steps simplifies the manufacturing workflow, reducing both labor hours and the consumption of auxiliary materials like silica gel or specialized resins.
- Cost Reduction in Manufacturing: The adoption of non-metallic catalysts eliminates the need for costly heavy metal removal processes, which are often required to meet strict pharmacopeial limits. By avoiding these downstream purification bottlenecks, manufacturers can achieve substantial cost savings in the overall production budget. Additionally, the catalyst loading is extremely low, further minimizing the expense associated with catalytic reagents while maintaining high turnover numbers.
- Enhanced Supply Chain Reliability: The robustness of the borane catalyst, which exhibits tolerance to moisture and air compared to sensitive transition metal complexes, ensures more consistent batch-to-batch performance. This reliability reduces the risk of production delays caused by catalyst deactivation or stringent environmental controls. Consequently, suppliers can offer more predictable lead times and maintain higher inventory turnover rates for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The one-pot nature of this reaction significantly reduces solvent usage and waste generation compared to multi-step metal-catalyzed sequences. This aligns with green chemistry principles and simplifies environmental compliance reporting. The mild reaction conditions also lower energy consumption, making the commercial scale-up of complex pharmaceutical intermediates more sustainable and economically viable for large-volume production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers.
Q: Why is metal-free catalysis critical for quinoline-4-carboxylate intermediates?
A: Traditional methods utilize heavy metal catalysts like Ytterbium or Iridium, which pose significant risks of residual contamination in final API products. The metal-free borane approach eliminates the need for expensive and complex metal scavenging steps, ensuring compliance with stringent pharmaceutical purity standards.
Q: What are the substrate scope capabilities of this borane-catalyzed reaction?
A: The patented method demonstrates exceptional versatility, accommodating a wide range of aniline derivatives with various substituents (fluoro, chloro, methoxy), aromatic or aliphatic aldehydes, and different pyruvate esters, delivering consistent yields up to 95%.
Q: How does this process impact commercial scalability and cost?
A: By operating under mild conditions (0-100°C) and using air- and moisture-tolerant catalysts, the process reduces energy consumption and simplifies operational requirements. The one-pot nature minimizes waste generation and processing time, leading to substantial cost savings in large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline-4-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to ensure the quality and availability of essential pharmaceutical intermediates. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the borane-catalyzed quinoline synthesis are translated into reliable supply. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for quinoline-based therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules, ensuring a seamless transition to this superior manufacturing technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
