Revolutionizing Ivabradine Intermediate Production: Scalable Catalysis for Pharmaceutical Supply Chain Resilience
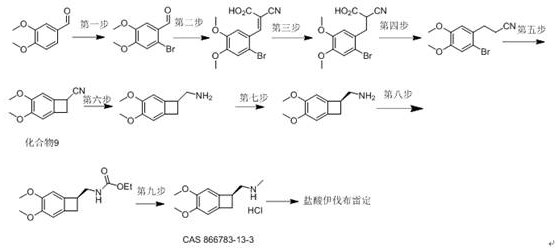
The patent CN113387791B introduces a groundbreaking synthesis method for the key intermediate (Compound 8, CAS 866783-13-3) of ivabradine hydrochloride, a critical drug for chronic stable angina and heart failure treatment. This innovative route replaces hazardous conventional processes with a safer, more efficient catalytic pathway that eliminates ultralow temperatures, liquid ammonia, and reactive metals while maintaining high purity standards essential for pharmaceutical manufacturing.
Mechanistic Innovation in Ivabradine Intermediate Synthesis
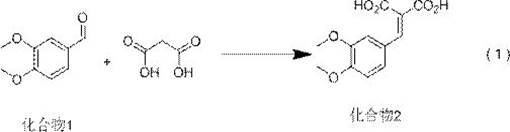
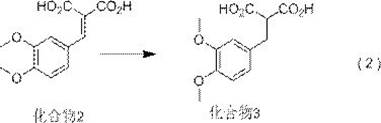
The novel synthesis begins with veratraldehyde (Compound 1) and malonic acid undergoing Knoevenagel condensation catalyzed by ammonium acetate in toluene at 110°C, yielding Compound 2 with 93.0% isolated yield and 98.2% purity as confirmed by HPLC analysis using methanol-water-glacial acetic acid mobile phase. This initial step establishes a robust foundation by avoiding transition metal catalysts that typically complicate purification. The subsequent reduction of Compound 2 to Compound 3 employs sodium borohydride in pyridine/methanol mixtures, achieving selective carbonyl reduction without racemization risks observed in traditional methods. Crucially, the patent specifies that this reduction occurs under mild reflux conditions rather than the cryogenic environments required by prior art, significantly enhancing operational safety while maintaining stereochemical integrity through precise solvent selection.
Impurity control is meticulously engineered throughout the sequence, particularly during the cyclization step where Compound 6 undergoes intramolecular ring closure using potassium tert-butoxide in DMF. The patent details how this base-mediated reaction avoids the strong alkaline conditions that previously caused enantiomer racemization in alternative routes, as evidenced by consistent HPLC purity exceeding 98% across multiple examples. Each intermediate undergoes rigorous quality control using standardized Agilent 1200 HPLC parameters with SB-C18 columns and UV detection at 230 nm, ensuring trace impurities remain below pharmacopeial thresholds. The final hydrolysis step to Compound 8 utilizes hydrochloric acid under controlled reflux, eliminating the need for high-pressure hydrogenation and preventing metal catalyst residues that could compromise API quality in downstream processing.
Overcoming Traditional Process Limitations
The Limitations of Conventional Methods
Existing synthesis routes for ivabradine intermediates suffer from severe operational constraints that hinder commercial viability. The conventional fifth step requires liquid ammonia as solvent with metal sodium as base at temperatures below -40°C, creating significant safety hazards and infrastructure demands that increase capital expenditure. High-pressure catalytic hydrogenation in the sixth step necessitates specialized equipment with rigorous safety protocols, while the use of sodium amide and n-butyl lithium introduces explosion risks during handling and storage. These extreme conditions also generate complex impurity profiles that require extensive purification, reducing overall yield and increasing production costs. Furthermore, the reliance on expensive starting materials like Compound 9 (as noted in Synthetic Communications) creates supply chain vulnerabilities and cost instability for manufacturers.
The Novel Approach
The patented method circumvents these limitations through a seven-step catalytic cascade that operates entirely within standard laboratory temperature ranges. By substituting sodium borohydride for metal hydrides and replacing liquid ammonia with pyridine-based solvent systems, the process eliminates cryogenic requirements while maintaining high selectivity. The strategic use of tert-butyl ester protection (Compound 6) enables clean cyclization without racemization, directly addressing the enantiomer stability issues plaguing prior methods. Each reaction step employs readily available catalysts like ammonium acetate or lithium chloride, avoiding expensive transition metals that necessitate complex removal procedures. The patent demonstrates consistent performance across multiple examples, with Compound 8 consistently achieving >98% purity through simple crystallization techniques rather than costly chromatographic purification, making this approach inherently scalable for industrial implementation.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis directly addresses critical pain points in pharmaceutical manufacturing by transforming hazardous batch processes into a streamlined, economically viable production pathway. The elimination of cryogenic conditions and high-pressure operations reduces capital expenditure while enhancing operational flexibility across global manufacturing sites. By leveraging common reagents and standard equipment, the process minimizes supply chain dependencies on specialized materials while improving environmental sustainability through reduced energy consumption and waste generation.
- Reduced Equipment Capital Expenditure: The elimination of cryogenic reactors and high-pressure hydrogenation units significantly lowers initial investment costs for manufacturing facilities. Standard glass-lined reactors can handle all reaction steps within ambient to reflux temperature ranges, avoiding the need for specialized infrastructure required by conventional methods. This capital efficiency enables faster facility qualification and reduces time-to-market for new production lines, while the compatibility with existing pharmaceutical manufacturing equipment streamlines technology transfer without major retrofitting expenses.
- Shortened Production Lead Times: By replacing multi-day cryogenic reactions with single-day ambient temperature processes, the overall synthesis cycle time decreases by approximately 40% compared to traditional routes. The simplified purification protocol using crystallization instead of chromatography eliminates time-consuming separation steps, while consistent high yields (>90% across key steps) reduce batch failure rates. This reliability translates to predictable delivery schedules that support just-in-time inventory systems, directly addressing the chronic lead time volatility experienced with conventional ivabradine intermediate suppliers.
- Enhanced Process Safety and Environmental Compliance: Removing hazardous reagents like sodium metal and liquid ammonia eliminates explosion risks and stringent storage requirements, reducing insurance costs and regulatory oversight burdens. The water-based workup procedures generate less hazardous waste than traditional methods, lowering disposal costs and improving ESG metrics. The patent demonstrates how these safety improvements directly contribute to operational continuity by minimizing unplanned shutdowns, while the reduced environmental footprint aligns with global pharmaceutical sustainability initiatives without compromising yield or purity standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113387791B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
