Scalable Synthesis of Erexib Intermediates for Commercial API Manufacturing
Scalable Synthesis of Erexib Intermediates for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for non-steroidal anti-inflammatory drugs (NSAIDs) that balance efficacy with production feasibility. Patent CN111747879A introduces a significant advancement in the large-process synthesis of Erexib, a potent COX-2 inhibitor used for relieving osteoarthritis pain. This technology addresses critical bottlenecks in traditional synthesis, such as reagent instability and harsh reaction conditions, by employing a novel sequence of acylation, reduction, and cyclization. The core innovation lies in the utilization of a stable amino-ketal intermediate, which circumvents the self-polymerization issues common in alpha-aminoketone chemistry. For global procurement teams and R&D directors, this represents a pivotal shift towards more reliable pharmaceutical intermediate supplier networks capable of delivering high-purity materials consistently.
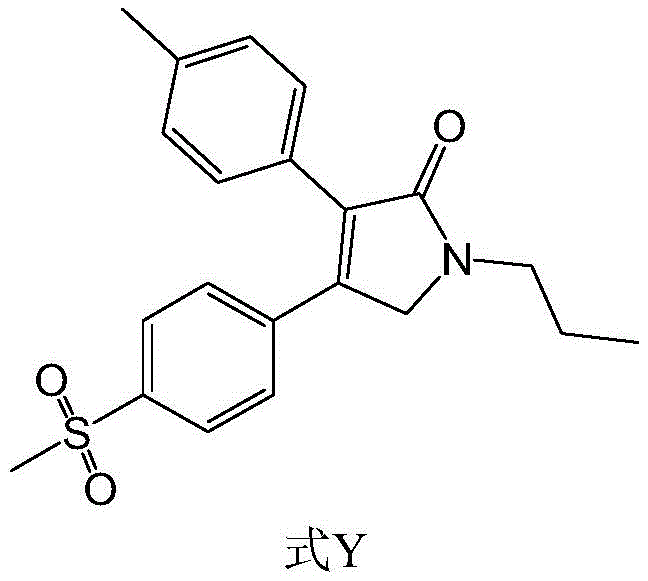
Erexib, chemically known as 1-n-propyl-3-(4-methylphenyl)-4-(4-methylsulfonylphenyl)-2,5-dihydro-1H-2-pyrrolidone, possesses a complex molecular architecture that demands precise stereochemical and functional group control. The structural integrity of the sulfone group and the pyrrolidone ring is paramount for biological activity. Traditional routes often struggle with the introduction of the N-propyl group and the formation of the five-membered lactam ring without generating significant impurities. The disclosed method in CN111747879A offers a streamlined approach that not only enhances the overall yield but also simplifies the downstream purification processes. By focusing on mild reagents and environmentally friendly solvents, this process aligns with modern green chemistry principles, reducing the environmental footprint associated with API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Erexib and its analogues has been plagued by the inherent instability of key precursors. Prior art, including patents like CN108707100A, often relies on alpha-aminoketones which are notoriously unstable under the alkaline conditions required for subsequent cyclization steps. This instability leads to rapid self-polymerization, resulting in tarry by-products that are extremely difficult to separate from the desired product. Furthermore, attempts to stabilize these intermediates as hydrochloride salts often necessitate the use of stronger bases in later stages, complicating the reaction profile and increasing the risk of side reactions. These factors contribute to low overall yields, high production costs, and significant challenges in waste management, making conventional routes less attractive for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
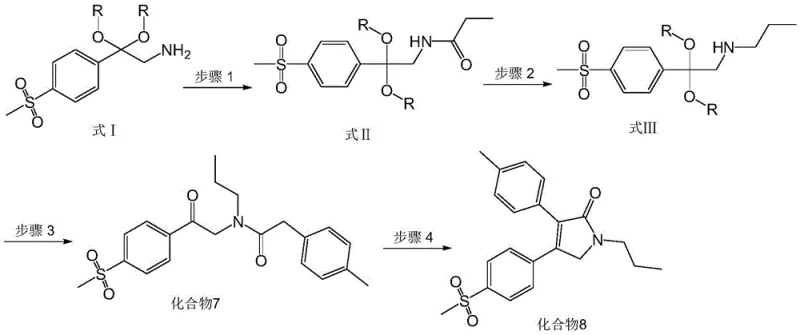
The methodology presented in CN111747879A fundamentally alters the synthetic landscape by introducing a stable ketal-amine building block (Compound 4) as the starting point for the final assembly. As illustrated in the reaction scheme below, the process proceeds through a logical sequence of acylation with propionyl chloride followed by a selective reduction using borane-THF. This strategy effectively bypasses the instability issues of free alpha-aminoketones. The subsequent condensation with p-methylphenylacetic acid and final base-mediated cyclization occur under mild conditions, utilizing common solvents like ethanol and acetonitrile. This novel approach not only improves the purity profile of the final API but also drastically simplifies the operational complexity, offering a clear pathway for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Amide Coupling and Cyclization
The success of this synthesis hinges on the precise control of the amide bond formation and the subsequent intramolecular cyclization. In Step 3, the condensation of the reduced amine (Compound 6) with p-methylphenylacetic acid is facilitated by a robust coupling system comprising EDCI, HOBt, and DMAP. This combination ensures high conversion rates while minimizing racemization or epimerization risks, which is critical for maintaining the pharmacological profile of the drug. The mechanistic pathway involves the activation of the carboxylic acid to form an active ester intermediate, which is then attacked by the nucleophilic amine. The presence of DMAP acts as a nucleophilic catalyst, accelerating the acylation process and ensuring that the reaction proceeds to completion even at room temperature, thereby preserving the integrity of other sensitive functional groups within the molecule.
Furthermore, the final cyclization step (Step 4) is a masterpiece of thermodynamic control. By employing potassium carbonate in an ethanol solution, the reaction creates a mildly basic environment that promotes the deprotonation of the amide nitrogen, initiating a nucleophilic attack on the adjacent ketone carbonyl (after ketal hydrolysis or direct displacement depending on the specific mechanistic nuance of the ketal stability). This intramolecular condensation closes the five-membered pyrrolidone ring efficiently. The choice of ethanol as a solvent is particularly strategic; it serves both as a reaction medium and potentially as a proton source/sink, facilitating the elimination of water or alcohol molecules during ring closure. This mechanistic elegance ensures that the final product, Erexib, is obtained with high purity, minimizing the need for extensive chromatographic purification which is often a bottleneck in high-purity API intermediate production.
How to Synthesize Erexib Efficiently
The synthesis of Erexib described in this patent is designed for operational simplicity and scalability. The process begins with the preparation of the critical intermediate, Compound 4, via a multi-step sequence involving oxime formation and rearrangement, ensuring a steady supply of high-quality starting material. Once Compound 4 is secured, the four-step transformation to the final API can be executed using standard reactor setups found in most fine chemical facilities. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature profiles, are outlined below to guide process engineers in replicating this high-yield route.
- React Compound 4 with propionyl chloride and triethylamine in acetonitrile to form the amide intermediate (Compound 5).
- Reduce Compound 5 using borane-tetrahydrofuran complex in THF under nitrogen protection to obtain the amine intermediate (Compound 6).
- Perform condensation of Compound 6 with p-methylphenylacetic acid using EDCI, HOBt, and DMAP to generate the linear precursor (Compound 7).
- Cyclize Compound 7 using potassium carbonate in ethanol solution under reflux to yield the final product, Erexib (Compound 8).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The shift away from unstable intermediates and harsh reagents translates directly into a more resilient supply chain. By utilizing reagents that are commodity chemicals rather than specialized, expensive catalysts, the process reduces dependency on single-source suppliers and mitigates the risk of raw material shortages. Additionally, the simplified purification protocols mean that production cycles can be shortened, allowing for faster turnaround times and improved responsiveness to market demand fluctuations. This operational agility is crucial for maintaining continuity in the supply of critical pain management medications.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the elimination of costly purification steps and the use of inexpensive, widely available reagents. Traditional methods often require expensive chromatography or multiple recrystallizations to remove polymeric by-products generated by unstable intermediates. In contrast, this novel route produces cleaner reaction mixtures, allowing for simpler isolation techniques like filtration and washing. Furthermore, the high yields reported in each step (e.g., 100% in Step 1, 79% in Step 4) maximize the throughput of raw materials, significantly lowering the cost of goods sold (COGS) and enhancing the overall profitability of Erexib manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures a consistent supply of high-quality intermediates. The stability of Compound 4 allows it to be stockpiled if necessary, providing a buffer against production disruptions. Moreover, the use of standard solvents like acetonitrile, THF, and ethanol means that solvent recovery and recycling systems can be easily integrated, further stabilizing operational costs. This reliability makes the manufacturer a preferred partner for long-term contracts, ensuring that downstream API producers can plan their production schedules with confidence and reduce lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process is inherently safer and cleaner. The avoidance of heavy metal catalysts and highly toxic reagents simplifies waste treatment and disposal, reducing the environmental compliance burden. The mild reaction conditions (mostly room temperature or moderate reflux) lower energy consumption compared to processes requiring cryogenic temperatures or extreme heat. These factors facilitate easier regulatory approval and support sustainable manufacturing practices, which are increasingly becoming a prerequisite for doing business with major multinational pharmaceutical companies focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Erexib. These answers are derived directly from the technical specifications and experimental data provided in Patent CN111747879A, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: Why is the new synthesis route for Erexib considered superior to conventional methods?
A: Conventional methods often rely on unstable alpha-aminoketones that self-polymerize under alkaline conditions, leading to low yields and difficult purification. The novel route utilizes a stable ketal-amine intermediate (Compound 4) and mild reaction conditions, significantly improving yield and process safety.
Q: What are the critical reagents used in the cyclization step?
A: The cyclization step (Step 4) utilizes potassium carbonate as the base in an ethanol solution. This mild basic environment facilitates the intramolecular condensation to form the pyrrolidone ring without degrading sensitive functional groups.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is designed for large-process synthesis. It employs common organic solvents like acetonitrile, THF, and ethanol, and avoids highly toxic or expensive reagents, making it economically viable and environmentally friendly for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erexib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN111747879A and is fully equipped to implement this advanced methodology. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to collaborate with us to leverage this innovative synthesis route for your Erexib requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and drive value for your organization.
