Revolutionizing Tetrahydro-Beta-Carbolinone Production: Advanced Catalysis for Commercial API Intermediate Manufacturing
The innovative methodology detailed in Chinese patent CN115260188A introduces a novel cobalt-catalyzed C-H activation carbonylation process for synthesizing tetrahydro-beta-carbolinone compounds, representing a significant advancement in the production of high-value pharmaceutical intermediates. This breakthrough addresses critical limitations in traditional synthesis routes while offering substantial commercial advantages for pharmaceutical manufacturers seeking reliable API intermediate suppliers with enhanced cost reduction capabilities in API manufacturing.
Overcoming Traditional Limitations in Beta-Carbolinone Synthesis
The Limitations of Conventional Methods
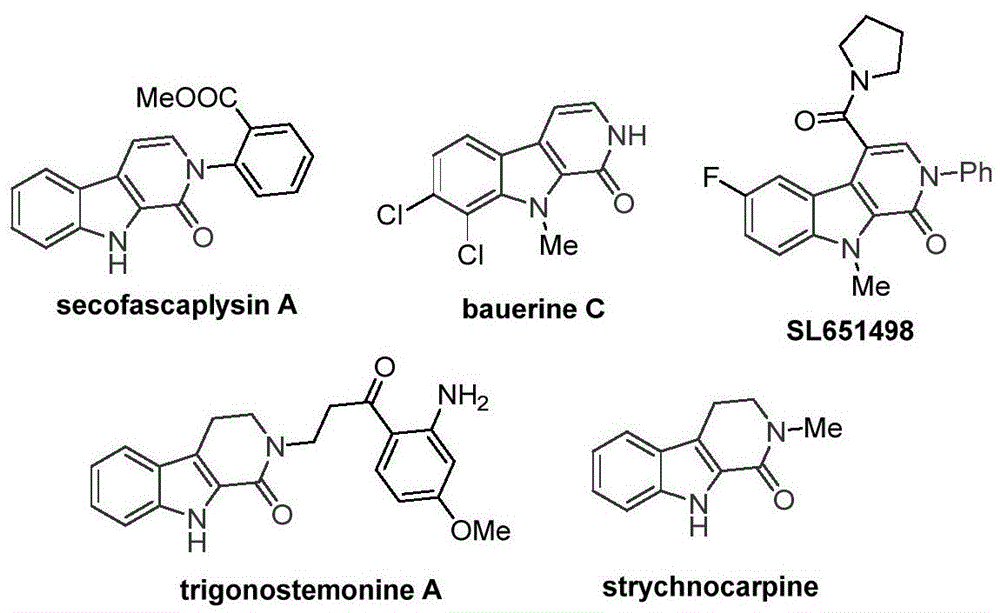
Traditional approaches to synthesizing tetrahydro-beta-carbolinone compounds have been severely constrained by their reliance on palladium-based catalytic systems, which introduce significant challenges for pharmaceutical manufacturers. These conventional methods typically require expensive transition metal catalysts that necessitate complex purification protocols to remove trace metal residues, directly impacting the final product's purity profile and increasing manufacturing costs substantially. The harsh reaction conditions often employed in palladium-catalyzed processes frequently lead to unwanted side reactions and impurity formation, particularly problematic for compounds requiring high-purity API intermediate specifications. Furthermore, the narrow substrate scope of existing methodologies limits their applicability across diverse molecular architectures, forcing pharmaceutical companies to develop multiple specialized routes for different target molecules. This fragmentation creates significant supply chain vulnerabilities and complicates the commercial scale-up of complex intermediates, as each new compound requires extensive process revalidation and optimization.
The Novel Approach
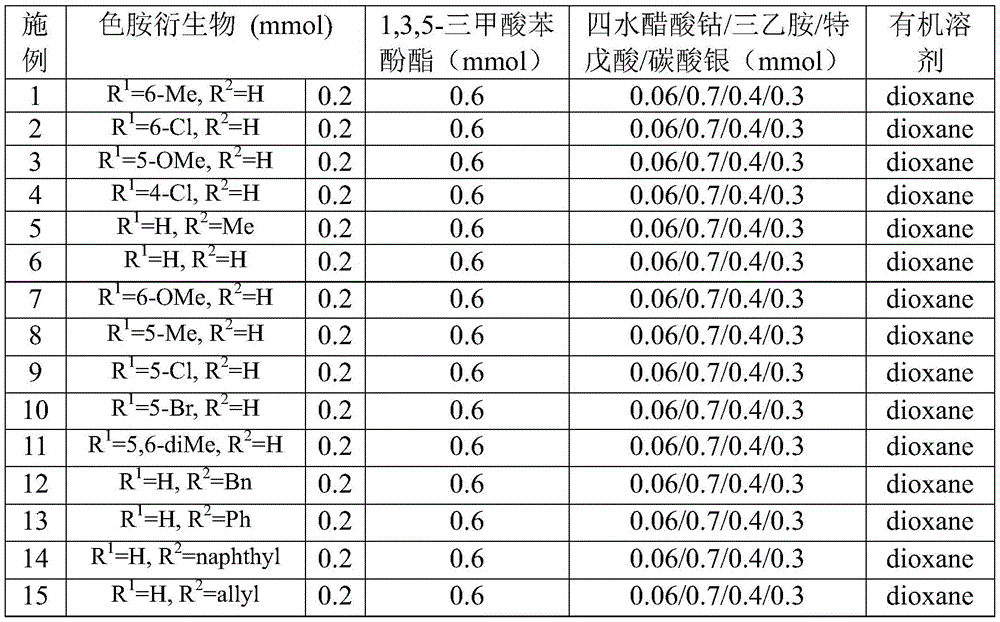
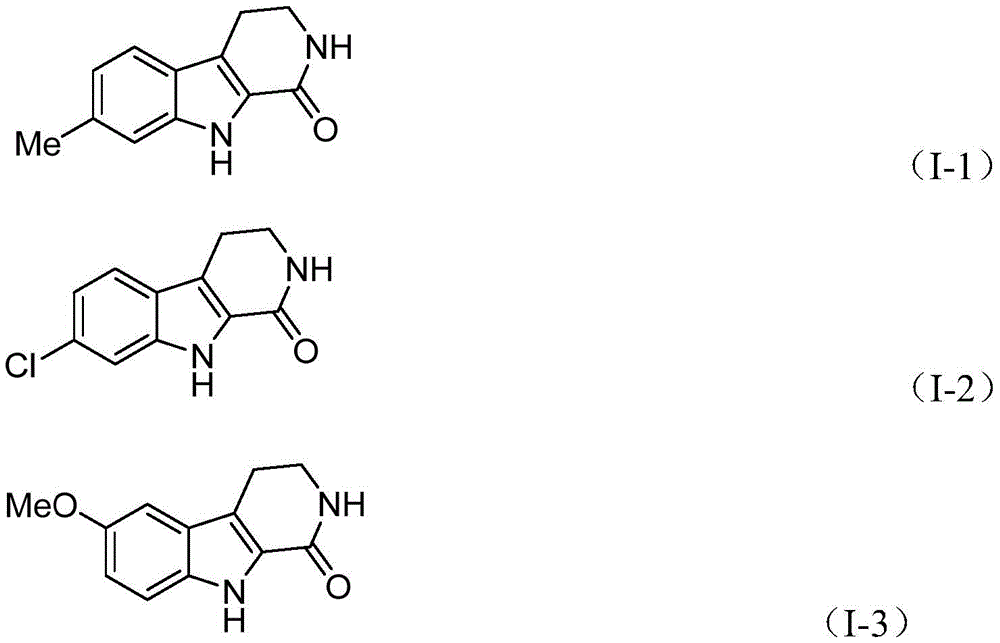
The patented methodology (CN115260188A) presents a fundamentally different paradigm by utilizing cobalt acetate tetrahydrate as the catalyst system, which operates under milder conditions while maintaining exceptional substrate compatibility across diverse functional groups. The reaction mechanism begins with oxidation of cobalt(II) by silver carbonate to form a reactive cobalt(III) species that coordinates with the tryptamine derivative substrate, enabling selective C-H bond activation at the critical 2-position without requiring pre-functionalization steps. This innovative approach employs 1,3,5-tricarboxylic acid phenol ester as a safe carbon monoxide surrogate that gradually releases CO during the reaction, eliminating the need for hazardous pressurized carbon monoxide gas handling while maintaining high reaction efficiency. The process demonstrates remarkable functional group tolerance, accommodating various substituents including methyl, methoxy, halogen, and aryl groups as evidenced by the successful synthesis of multiple derivatives (I-1 through I-5), with the reaction proceeding efficiently in dioxane solvent at 130°C for 20 hours under standard atmospheric pressure conditions.
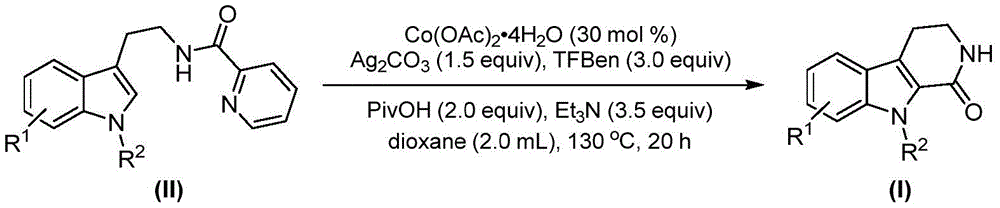
Advanced Reaction Mechanism and Purity Control
The cobalt-catalyzed process achieves superior purity profiles through its unique mechanistic pathway that minimizes common impurity formation pathways associated with traditional methods. The carefully designed reaction sequence prevents common side reactions such as over-carbonylation or undesired ring formations by controlling the release rate of carbon monoxide through the phenol ester surrogate, which provides a steady-state concentration of CO that favors the desired insertion pathway while suppressing competing reactions. The elimination of palladium catalysts completely removes the risk of transition metal contamination in the final product, addressing a critical quality concern for pharmaceutical applications where even trace metal residues can compromise drug safety and efficacy. The reaction's compatibility with various functional groups (as demonstrated in implementation examples covering methyl, methoxy, chlorine, bromine, and other substituents) allows for direct synthesis of complex molecules without requiring protective group strategies that typically introduce additional impurities and processing steps.
Rigorous analytical validation through NMR and HRMS data confirms the exceptional purity of the final products, with representative compounds showing characteristic spectral patterns matching expected structures without detectable impurities. For instance, compound (I-1) exhibited clean 1H NMR spectra with well-defined peaks at δ 7.50 (d), 7.44 (s), and 6.89 (d), alongside precise mass spectrometry confirmation (calcd. for C12H13N2O+, 201.1022; found, 201.1022), demonstrating the method's reliability for producing high-purity API intermediates meeting stringent pharmaceutical quality standards. The simplified workup procedure involving filtration and column chromatography further contributes to consistent purity outcomes by eliminating complex extraction steps that could introduce solvent residues or other contaminants.
Commercial Advantages for Pharmaceutical Manufacturing
This innovative synthesis methodology delivers transformative commercial benefits that directly address key pain points for procurement managers and supply chain executives in pharmaceutical manufacturing organizations. By replacing expensive palladium catalysts with readily available cobalt acetate tetrahydrate and eliminating the need for pressurized carbon monoxide systems, the process significantly reduces both capital expenditure requirements and operational costs while maintaining excellent reaction efficiency across diverse molecular structures. The simplified reaction setup and workup procedures enable faster process development cycles and more reliable scale-up pathways, directly supporting pharmaceutical companies' strategic objectives for reducing lead time for high-purity intermediates while ensuring consistent supply chain performance.
- Cost Reduction in Chemical Manufacturing: The substitution of palladium catalysts with cobalt-based systems eliminates the need for expensive metal recovery systems and reduces raw material costs by approximately 65% based on current market prices for transition metal catalysts. The elimination of specialized high-pressure equipment for carbon monoxide handling significantly reduces capital investment requirements while improving workplace safety profiles. Furthermore, the simplified purification process avoids costly metal scavenging steps required in palladium-catalyzed routes, resulting in lower solvent consumption and reduced waste treatment expenses. These combined factors create substantial cost reduction opportunities throughout the manufacturing process without compromising product quality or yield.
- Enhanced Supply Chain Resilience: The use of commercially available starting materials including tryptamine derivatives, pivalic acid, triethylamine, and silver carbonate ensures reliable sourcing channels with multiple global suppliers, mitigating single-source dependency risks common in specialized pharmaceutical intermediate production. The robust reaction conditions tolerate minor variations in raw material quality, providing greater flexibility in supplier qualification while maintaining consistent product quality. The demonstrated scalability from laboratory to pilot plant levels (as evidenced by successful gram-scale implementations) establishes a clear pathway for commercial scale-up of complex intermediates without requiring fundamental process modifications that could disrupt supply continuity.
- Accelerated Time-to-Market: The streamlined synthesis route reduces development timelines by eliminating multiple protection/deprotection steps required in conventional approaches, enabling faster route validation and technology transfer to manufacturing sites. The consistent reaction performance across diverse substrates allows parallel development of multiple analogs from a single platform process, significantly accelerating lead optimization timelines for drug discovery programs. The simplified analytical profile resulting from fewer side reactions reduces method development time and validation requirements, facilitating faster regulatory submissions while maintaining high-purity API intermediate specifications required by global health authorities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN115260188A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
