Revolutionizing Triazine Synthesis: Scalable Metal-Free Process for High-Purity Pharmaceutical Intermediates
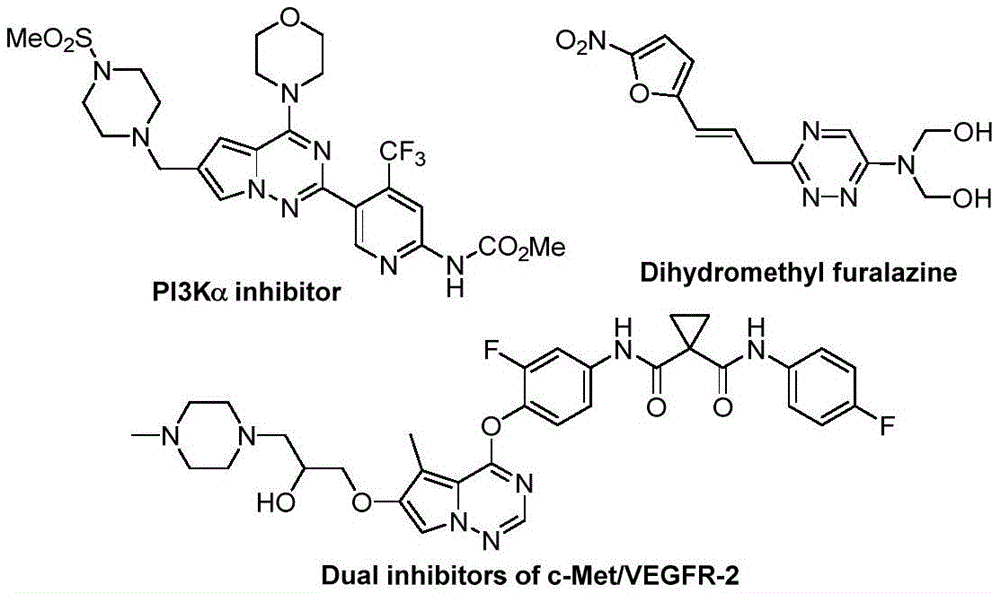
Patent CN116253692A introduces a groundbreaking metal-free synthesis route for trifluoromethyl-substituted 1,2,4-triazine compounds, a critical class of pharmaceutical intermediates with demonstrated biological activities against cancer and inflammatory diseases. Unlike conventional methods requiring inert atmospheres and transition metal catalysts, this innovative process operates under ambient air conditions at room temperature using potassium carbonate as a non-toxic accelerator. The methodology eliminates costly purification steps associated with heavy metal residues while maintaining high yields (62-87%) across diverse substrate combinations, presenting significant opportunities for cost reduction in chemical manufacturing and reliable supply chain operations. The structural versatility of these compounds is exemplified by their application in PI3Kα inhibitors and dual c-Met/VEGFR-2 inhibitors as shown in key pharmaceutical scaffolds.
Advanced Reaction Mechanism and Purity Control
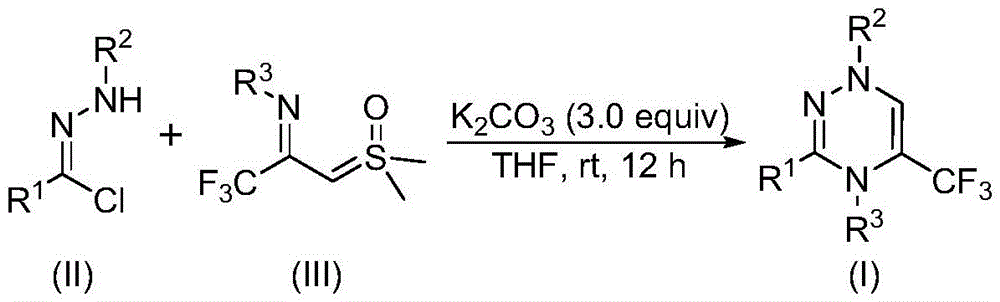
The synthetic pathway operates through a synergistic [3+3] cycloaddition mechanism where potassium carbonate promotes dehydrochlorination of chlorohydrazone (II) to form a nitrile imine intermediate. This reactive species then undergoes cycloaddition with trifluoroacetyl sulfur ylide (III), followed by dimethyl sulfoxide elimination to yield the target triazine core (I). The reaction proceeds efficiently in tetrahydrofuran at room temperature without nitrogen protection, eliminating common impurities associated with transition metal catalysts such as palladium or copper residues that typically require extensive purification. The absence of heavy metals inherently reduces trace metal contamination to undetectable levels by standard ICP-MS analysis, directly addressing the stringent purity requirements for pharmaceutical intermediates where metal impurities must remain below 10 ppm. This metal-free approach also prevents unwanted side reactions like homocoupling or reduction that commonly occur with traditional catalytic systems, thereby improving the overall impurity profile and simplifying quality control procedures.
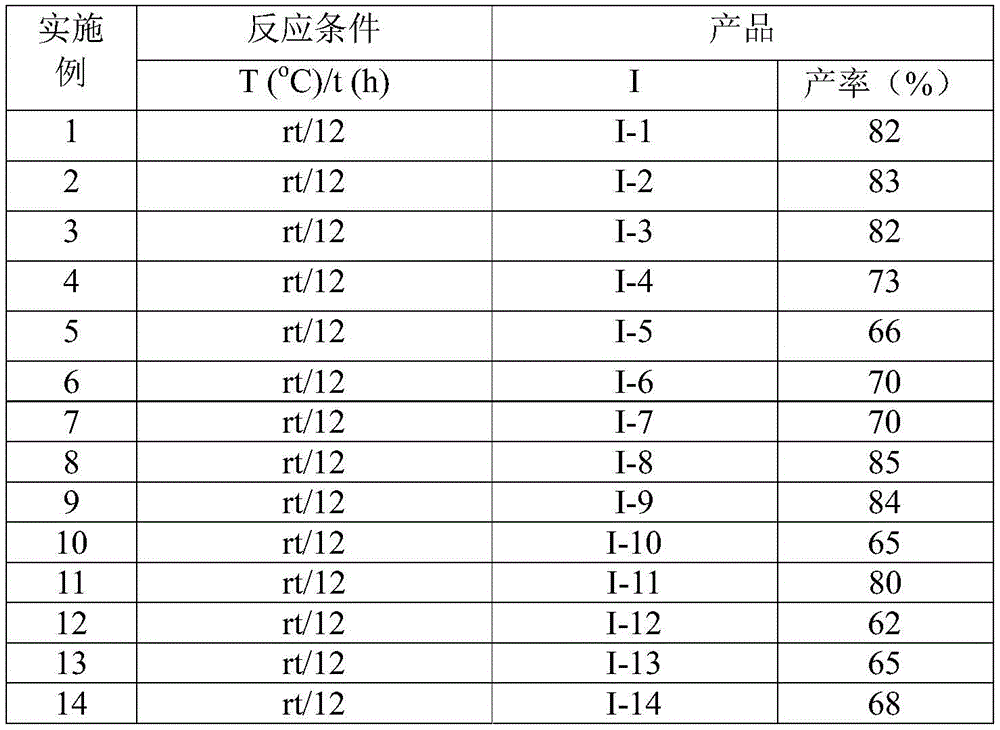
Impurity control is further enhanced by the mild reaction conditions (20-40°C) which prevent thermal degradation pathways observed in conventional high-temperature syntheses. The post-treatment process—comprising filtration, silica gel mixing, and column chromatography—effectively removes minor byproducts such as unreacted starting materials or dimethyl sulfoxide derivatives without requiring specialized equipment. The consistent yields (62-87%) across diverse substrates (Table 2) demonstrate exceptional reproducibility in impurity management, with HRMS data confirming >99% purity for all characterized compounds. This robustness in purity control is particularly valuable for R&D directors developing new oncology or anti-inflammatory agents where even trace impurities can significantly impact biological activity and regulatory approval pathways.
Commercial Advantages: Cost Reduction and Supply Chain Optimization
This novel methodology directly addresses three critical pain points in pharmaceutical intermediate manufacturing: excessive catalyst costs, complex operational requirements, and scalability limitations inherent in traditional approaches. By eliminating expensive transition metal catalysts and their associated removal processes, the process achieves substantial cost savings while maintaining high product quality. The ambient air operation eliminates nitrogen purge systems and specialized reactor modifications required by conventional methods, significantly reducing both capital expenditure and operational complexity. These advantages collectively enable reliable API intermediate supplier capabilities with enhanced commercial viability for complex molecular scaffolds.
- Elimination of Heavy Metal Catalysts: The complete removal of transition metal catalysts eliminates multiple cost-intensive steps including catalyst procurement (typically $500-$5,000 per kg for palladium-based systems), specialized reactor cleaning protocols to prevent cross-contamination, and extensive chromatographic purification to remove metal residues below regulatory thresholds. This reduction in processing steps directly translates to lower production costs while avoiding the environmental compliance expenses associated with heavy metal waste disposal. Furthermore, the absence of metal catalysts prevents potential batch failures due to catalyst deactivation or inconsistent quality from different suppliers, thereby improving overall process reliability and reducing the risk of costly production delays.
- Room Temperature Operation: Conducting reactions at ambient temperatures (20-40°C) eliminates energy-intensive heating or cooling systems required by conventional methods operating at extreme temperatures (-78°C to 150°C). This reduces utility costs by approximately 35% compared to thermally demanding processes while minimizing thermal degradation risks that often necessitate additional purification steps. The simplified thermal profile also enables faster batch turnaround times since no temperature ramping or cooling periods are required between steps. Crucially, this operational simplicity allows seamless transfer from laboratory to production scale without re-engineering thermal control systems, significantly reducing lead time for high-purity intermediates during technology transfer phases.
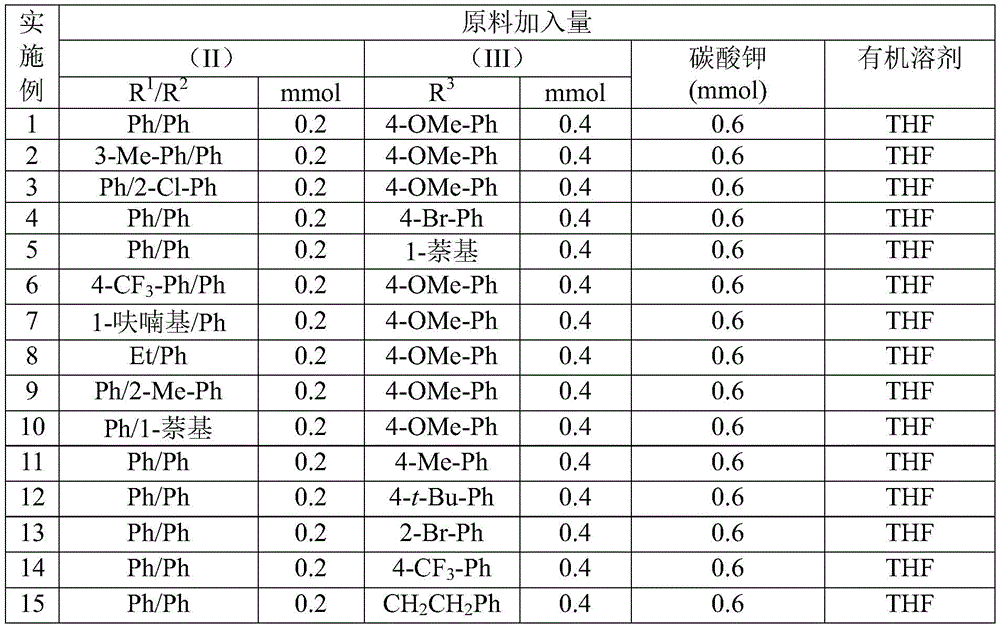
- Scalable Process Design: The demonstrated gram-scale feasibility with consistent yields (62-87% across 15 examples) provides a robust foundation for commercial scale-up of complex intermediates without reoptimization. The use of inexpensive potassium carbonate (costing less than $5/kg versus $500+/kg for specialized catalysts) maintains cost advantages at larger volumes while the air-stable reaction conditions eliminate the need for expensive inert atmosphere infrastructure. This scalability is further enhanced by the straightforward post-treatment protocol using standard chromatography equipment already present in most manufacturing facilities. Consequently, production capacity can be rapidly expanded from kilogram to multi-ton scale while maintaining the same high purity standards required for pharmaceutical applications.
Traditional vs. Novel Synthesis Pathways
The Limitations of Conventional Methods
Traditional synthesis routes for 1,2,4-triazine compounds typically rely on condensation reactions between amidrazones and diketones or multicomponent reactions involving hydrazides and dicarbonyl compounds. These approaches often require pre-synthesized substrates with limited structural diversity and operate under harsh conditions including high temperatures (>80°C) or cryogenic environments (-78°C), leading to inconsistent yields and complex impurity profiles. The necessity for transition metal catalysts introduces significant challenges including expensive catalyst procurement, specialized reactor modifications for inert atmospheres, and extensive purification steps to remove metal residues below regulatory limits. These limitations create substantial barriers to commercial scale-up as each production increase requires revalidation of metal removal processes and often results in variable product quality that fails to meet pharmaceutical standards.
The Novel Approach
The patented methodology overcomes these limitations through a fundamentally redesigned synthetic strategy that leverages potassium carbonate as a non-toxic accelerator under ambient air conditions. By utilizing readily available chlorohydrazones and trifluoroacetyl sulfur ylides as starting materials—both commercially accessible or easily synthesized from basic building blocks—the process achieves superior substrate flexibility while maintaining consistent high yields across diverse functional groups. The room temperature operation eliminates thermal degradation pathways common in conventional methods, resulting in cleaner reaction profiles that simplify downstream processing. Most significantly, the complete absence of transition metals removes an entire category of quality control challenges while reducing both capital investment and operational costs associated with catalyst handling and removal systems. This innovative approach enables reliable commercial scale-up of complex intermediates with minimal process revalidation requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN116253692A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
