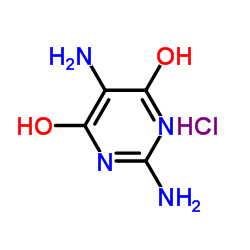
2,5-Diamino-4,6-dihydroxypyrimidine Hydrochloride
- CAS No.56830-58-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 2,5-Diamino-4,6-dihydroxypyrimidine Hydrochloride for pharmaceutical synthesis. Reliable supply with full documentation and COA.
Request Bulk PricingProduct Technical Details
Product Overview
2,5-Diamino-4,6-dihydroxypyrimidine Hydrochloride is a highly specialized heterocyclic compound serving as a critical building block in modern medicinal chemistry. This advanced intermediate is characterized by its stable pyrimidine ring structure, making it indispensable for the synthesis of complex nucleoside analogues. Our manufacturing process ensures exceptional purity levels, catering to the rigorous demands of pharmaceutical research and development laboratories worldwide. As a key precursor, it facilitates the efficient construction of antiviral agents, supporting the global supply chain for essential therapeutic interventions.
Manufactured under strict quality control protocols, this chemical exhibits consistent physicochemical properties essential for reproducible reaction outcomes. The hydrochloride salt form enhances solubility and stability during various synthetic transformations. We prioritize safety and compliance in every batch, providing comprehensive documentation to support regulatory submissions and internal quality audits.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 56830-58-1 |
| Molecular Formula | C4H7ClN4O2 |
| Molecular Weight | 178.577 g/mol |
| Appearance | Light yellow or light red crystalline powder |
| Assay | ≥98.5% |
| Moisture Content | ≤0.5% |
| Boiling Point | 490ºC at 760 mmHg |
| Flash Point | 250.1ºC |
Industrial Applications
This compound is primarily utilized in the pharmaceutical sector as a vital intermediate for the synthesis of antiretroviral medications. Its structural configuration allows for specific functionalization required in the production of nucleoside reverse transcriptase inhibitors. Researchers rely on this high-purity material to optimize reaction yields and minimize impurity profiles in downstream processing. Beyond specific antiviral applications, it serves as a valuable tool in organic synthesis for developing novel heterocyclic libraries.
Our clients leverage this intermediate for scale-up processes, ensuring that pilot plant operations transition smoothly to commercial manufacturing. The consistency of our product reduces variability in final API production, contributing to cost-effective and reliable pharmaceutical manufacturing workflows.
Quality Assurance and Storage
Every production batch undergoes rigorous analytical testing using high-performance liquid chromatography and nuclear magnetic resonance spectroscopy. We provide a Certificate of Analysis (COA) with each shipment, detailing all critical quality attributes. To maintain integrity, the product should be stored in a cool, dry, and well-ventilated area away from direct sunlight and incompatible materials. Proper sealing is recommended to prevent moisture absorption, which could affect performance in moisture-sensitive reactions.
Handling this chemical requires standard laboratory safety precautions. Personnel should wear appropriate personal protective equipment, including chemical-resistant gloves and safety goggles, to prevent skin and eye contact. In case of accidental exposure, immediate washing with plenty of water is advised. The material is stable under recommended storage conditions but should be kept away from strong oxidizing agents. Disposal of waste material must comply with local environmental regulations to ensure ecological safety.
We are committed to delivering excellence in chemical supply. Our logistics network ensures timely global delivery while maintaining compliance with international shipping regulations for chemical substances. For technical inquiries or custom packaging requirements, our expert support team is available to assist with your specific project needs.



