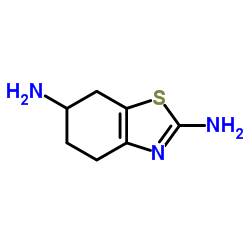
2,6-Diamino-4,5,6,7-tetrahydrobenzothiazole
- CAS No.104617-49-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate essential for Pramipexole synthesis, offering ≥99% purity and global supply reliability.
Request Bulk PricingProduct Technical Details
Product Overview
2,6-Diamino-4,5,6,7-tetrahydrobenzothiazole is a critical pharmaceutical intermediate utilized extensively in the synthesis of advanced therapeutic agents. This compound serves as a foundational building block in the manufacturing process of Pramipexole, a well-known medication used in the management of neurological conditions. Our facility specializes in the production of this high-value chemical entity, adhering to strict quality control protocols to ensure consistency and reliability for downstream pharmaceutical applications.
As a dedicated manufacturer, we understand the importance of molecular integrity in drug synthesis. Our production lines are optimized to deliver material with exceptional purity levels, minimizing impurities that could affect subsequent reaction steps. This commitment to quality makes our product a preferred choice for research laboratories and large-scale API manufacturing plants globally.
Technical Specifications
We maintain rigorous testing standards for every batch produced. The following table outlines the key physical and chemical parameters guaranteed for this product:
| Parameter | Specification |
|---|---|
| CAS Number | 104617-49-4 |
| Molecular Formula | C7H11N3S |
| Molecular Weight | 169.25 g/mol |
| Appearance | Almost white or light gray powder |
| Purity (HPLC) | ≥99.0% |
| Related Substances | ≤0.5% |
| Residue on Ignition | ≤0.5% |
| Heavy Metals | ≤20 ppm |
| Water Content | ≤1.0% |
| Density | 1.313 g/cm³ |
| Boiling Point | 359°C at 760 mmHg |
| Flash Point | 170.9°C |
Industrial Applications
The primary application of 2,6-Diamino-4,5,6,7-tetrahydrobenzothiazole lies within the pharmaceutical sector, specifically as a key intermediate in the multi-step synthesis of Pramipexole dihydrochloride. Its unique chemical structure facilitates specific coupling reactions required to construct the final active pharmaceutical ingredient (API). Beyond this primary use, it serves as a valuable reagent in organic synthesis research, enabling chemists to explore new heterocyclic compounds and potential therapeutic candidates.
Our clients utilize this material for both process development and commercial-scale production. The high purity ensures that downstream purification processes are more efficient, reducing overall production costs and time-to-market for finished pharmaceutical products.
Quality Assurance and Packaging
Quality is paramount in our manufacturing process. Each batch undergoes comprehensive analysis using advanced chromatographic and spectroscopic methods. A Certificate of Analysis (COA) is provided with every shipment, detailing the test results against our internal specifications and international standards. We operate under GMP-like conditions to ensure product safety and traceability.
Standard packaging consists of 25kg cardboard drums, designed to protect the material from moisture and light during transit. Custom packaging solutions are available upon request to meet specific logistical requirements. For storage, we recommend keeping the container tightly closed in a cool, dry, and well-ventilated area, away from strong oxidizing agents and direct sunlight.
Why Choose Our Supply
- Consistent high purity ≥99% for reliable synthesis outcomes.
- Robust supply chain capable of meeting global demand.
- Comprehensive technical support and documentation.
- Competitive bulk pricing for long-term partnerships.
- Strict compliance with international chemical safety regulations.
Contact our sales team today to request a sample or discuss your specific volume requirements. We are committed to supporting your research and production goals with premium chemical intermediates.



