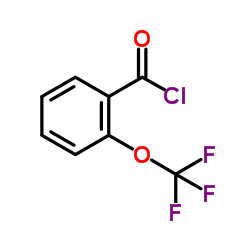
2-(Trifluoromethoxy)benzoyl Chloride
- CAS No.162046-61-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 2-(Trifluoromethoxy)benzoyl chloride designed for advanced organic synthesis and pharmaceutical intermediate production with reliable quality assurance.
Request Bulk PricingProduct Technical Details
Product Overview
2-(Trifluoromethoxy)benzoyl Chloride is a specialized fluorinated aromatic acyl chloride utilized extensively in the field of fine chemical synthesis and pharmaceutical development. As a key building block, this compound introduces the trifluoromethoxy functional group into complex molecular structures, enhancing metabolic stability and lipophilicity in final active pharmaceutical ingredients. Our manufacturing process ensures consistent quality and high purity levels suitable for demanding synthetic routes.
The presence of the trifluoromethoxy moiety offers distinct advantages in medicinal chemistry, often improving the bioavailability and binding affinity of drug candidates. This makes the compound an essential reagent for researchers and industrial chemists working on kinase inhibitors, agrochemicals, and advanced material science projects. We maintain strict control over production parameters to minimize impurities and ensure batch-to-batch reproducibility.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 2-(Trifluoromethoxy)benzoyl Chloride |
| CAS Number | 162046-61-9 |
| Molecular Formula | C8H4ClF3O2 |
| Molecular Weight | 224.56 g/mol |
| Appearance | Yellow Transparent Liquid |
| Assay (Purity) | ≥98.0% |
| Moisture Content | ≤0.2% |
| Density | 1.44 g/cm3 |
| Boiling Point | 94°C |
| Refractive Index | 1.47 |
Industrial Applications
This fluorinated intermediate serves as a critical precursor in multi-step organic synthesis. It is primarily employed in the acylation of amines, alcohols, and other nucleophiles to construct amide or ester linkages within larger molecular frameworks. The electron-withdrawing nature of the trifluoromethoxy group influences the reactivity of the carbonyl carbon, allowing for controlled reaction kinetics under various conditions.
Common applications include the synthesis of pharmaceutical APIs where fluorine substitution is required to modulate pharmacokinetic properties. Additionally, it finds use in the production of agrochemicals and specialty polymers. Our technical team supports customers with guidance on optimal reaction conditions, solvent selection, and safety protocols to maximize yield and efficiency in downstream processes.
Quality Assurance and Safety
Quality is paramount in our production facility. Every batch undergoes rigorous testing using advanced analytical techniques such as GC, HPLC, and NMR to verify identity and purity. We provide comprehensive Certificates of Analysis (COA) with each shipment, detailing all relevant quality parameters. Our facility adheres to international safety standards, ensuring that handling procedures minimize exposure risks associated with acyl chlorides.
Proper storage is essential to maintain product stability. The compound should be kept in a cool, ventilated area away from moisture and incompatible materials such as strong oxidizing agents or bases. We recommend using dry inert atmospheres during handling to prevent hydrolysis. Our packaging solutions are designed to protect the integrity of the chemical during transit, ensuring it arrives in optimal condition for immediate use in your laboratory or production line.
Packaging and Logistics
We offer flexible packaging options to meet diverse customer needs. Standard packaging includes 200 kg drums, which are securely sealed to prevent leakage and contamination. Custom packaging configurations are available upon request to accommodate specific logistical requirements or smaller scale research needs. Our global shipping network ensures timely delivery to major chemical hubs worldwide, supported by full documentation for customs and regulatory compliance.



