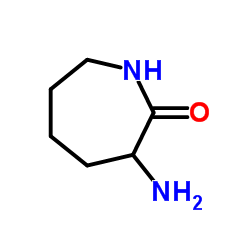
(3R)-3-aminoazepan-2-one
- CAS No.28957-33-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity (3R)-3-aminoazepan-2-one is a critical chiral intermediate for fluoroquinolone antibiotics. We offer reliable bulk supply with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
(3R)-3-aminoazepan-2-one, also known commercially as D-2-aminohexano-6-lactam, represents a highly specialized chiral building block essential for the synthesis of advanced pharmaceutical compounds. This seven-membered lactam structure features a critical stereocenter at the 3-position, making it indispensable for producing enantiomerically pure active pharmaceutical ingredients. Our manufacturing process ensures exceptional optical purity, catering specifically to the rigorous demands of modern medicinal chemistry and large-scale drug production.
As a leading global manufacturer, we understand the pivotal role this intermediate plays in the development of next-generation antibiotics. The compound serves as a foundational scaffold for fluoroquinolone derivatives, requiring precise control over stereochemistry to maintain biological efficacy. Our facility employs state-of-the-art asymmetric synthesis and purification technologies to guarantee consistent batch-to-bquality.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | (3R)-3-aminoazepan-2-one |
| CAS Registry Number | 28957-33-7 |
| Molecular Formula | C6H12N2O |
| Molecular Weight | 128.17 g/mol |
| Appearance | Yellow crystals |
| Assay (Purity) | ≥97.0% |
| Enantiomeric Excess (EE) | ≥98.0% |
| Density | 0.893 g/cm3 |
| Boiling Point | 180.9°C at 760 mmHg |
| Flash Point | 69.6°C |
Industrial Applications
This chiral lactam is primarily utilized as a key intermediate in the synthesis of potent fluoroquinolone antibiotics. Specifically, it is employed in the manufacturing processes of Balofloxacin and Besifloxacin hydrochloride. The high enantiomeric excess provided by our product ensures that the final drug substances meet stringent pharmacopoeial standards for optical purity, which is critical for safety and efficacy profiles.
Beyond antibiotic synthesis, this compound serves as a valuable building block for research and development in neurological therapeutics and other complex organic synthesis pathways. Pharmaceutical companies rely on this intermediate to streamline their production routes, reducing waste and improving overall yield in multi-step synthesis sequences.
Quality Assurance and Safety
Quality control is paramount in our production facility. Every batch of (3R)-3-aminoazepan-2-one undergoes rigorous testing using high-performance liquid chromatography (HPLC) and gas chromatography (GC) to verify purity and stereochemical integrity. We provide a comprehensive Certificate of Analysis (COA) with every shipment, detailing all physical and chemical parameters.
- Strict adherence to ISO quality management systems.
- Advanced analytical verification for impurities and residual solvents.
- Stable supply chain ensuring timely delivery for global clients.
Regarding safety, this chemical should be handled in accordance with standard industrial hygiene practices. It must be stored in a cool, dry, and well-ventilated area, kept away from moisture and strong light or heat sources. Proper personal protective equipment, including gloves and safety goggles, is recommended during handling to prevent irritation.
Packaging and Logistics
We offer flexible packaging solutions to meet diverse client needs. The standard packaging configuration is 25kg cardboard drums, which ensure product integrity during transit. Custom packaging options are available upon request to accommodate specific logistical requirements. Our logistics team coordinates efficiently to ensure secure and timely global delivery, maintaining the cold chain or specific storage conditions if necessary.



