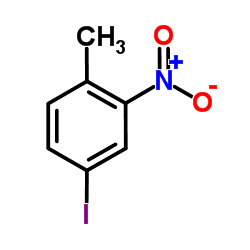
4-Iodo-2-nitrotoluene
- CAS No.41252-97-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 4-Iodo-2-nitrotoluene (CAS 41252-97-5) designed for advanced organic synthesis. Reliable supply for pharmaceutical and chemical manufacturing processes.
Request Bulk PricingProduct Technical Details
Product Overview
4-Iodo-2-nitrotoluene represents a vital class of halogenated nitroaromatic compounds utilized extensively in modern chemical synthesis. This specialized intermediate features a unique molecular structure incorporating both an iodine substituent and a nitro group on a toluene backbone. The presence of the iodine atom provides exceptional reactivity for cross-coupling reactions, while the nitro group offers versatile pathways for reduction and functionalization. Our manufacturing process ensures consistent quality suitable for demanding research and industrial applications.
As a key building block, this compound supports the development of complex organic molecules. The high level of purity achieved through our refined production methods minimizes impurities that could interfere with downstream reactions. We prioritize strict quality control measures at every stage of production to deliver a product that meets rigorous international standards for chemical intermediates.
Technical Specifications
The following table outlines the critical physical and chemical properties verified through our quality assurance laboratory. Each batch is tested to ensure compliance with the specified parameters.
| Parameter | Value |
|---|---|
| Chemical Name | 4-Iodo-2-nitrotoluene |
| CAS Number | 41252-97-5 |
| Molecular Formula | C7H6INO2 |
| Molecular Weight | 263.033 g/mol |
| Appearance | Brown-yellow crystalline powder |
| Assay (Purity) | ≥97.0% |
| Density | 1.9±0.1 g/cm3 |
| Boiling Point | 286.9±20.0 °C at 760 mmHg |
| Melting Point | 43 °C |
| Flash Point | 127.3±21.8 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.644 |
Industrial Applications
This compound serves as a critical building block in the synthesis of pharmaceuticals, agrochemicals, and advanced materials. The aryl iodide functionality is particularly valuable in palladium-catalyzed coupling reactions such as Suzuki, Heck, and Sonogashira couplings. These reactions are fundamental in constructing carbon-carbon bonds required for complex molecule assembly. Additionally, the nitro group can be selectively reduced to an amine, providing further opportunities for derivatization in multi-step synthesis routes.
Chemical manufacturers rely on this intermediate for producing high-value specialty chemicals. Its stability and reactivity profile make it a preferred choice for process chemistry teams aiming to optimize yield and efficiency. Whether used in laboratory-scale research or large-scale production, the consistent quality ensures reproducible results across different batches.
Storage and Handling
To maintain stability and safety, proper storage conditions are essential. The product should be stored in a cool, ventilated place away from direct sunlight and heat sources. Containers must be kept tightly closed when not in use to prevent moisture absorption and contamination. Handling should be performed in a well-ventilated area with appropriate personal protective equipment, including gloves and safety goggles.
We offer flexible packaging options to suit various logistical requirements, including standard 25 kg drums. Custom packaging is available upon request to accommodate specific supply chain needs. Our global distribution network ensures timely delivery while maintaining product integrity during transit. For detailed safety data and regulatory compliance information, please refer to the provided Certificate of Analysis and Safety Data Sheet with each shipment.



