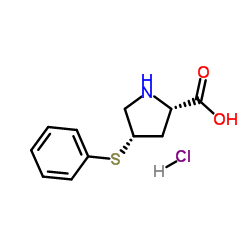
(4S)-4-(Phenylthio)-L-proline Hydrochloride
- CAS No.105107-84-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity (4S)-4-(Phenylthio)-L-proline Hydrochloride for Zofenopril synthesis. ≥98% assay, global shipping, and full COA documentation available for bulk orders.
Request Bulk PricingProduct Technical Details
Product Overview
(4S)-4-(Phenylthio)-L-proline Hydrochloride represents a critical chiral building block within the pharmaceutical industry, specifically designed for the synthesis of advanced cardiovascular therapeutics. As a specialized proline derivative, this compound exhibits unique stereochemical properties that are essential for ensuring the biological efficacy of downstream active pharmaceutical ingredients. Our manufacturing process adheres to strict quality control protocols to deliver consistent molecular integrity. This intermediate is widely recognized for its role in constructing complex peptide mimics and sulfur-containing drug molecules.
Chemical Properties
Chemically, this substance is characterized by the molecular formula C11H14ClNO2S and a molecular weight of 259.752 g/mol. The presence of the phenylthio group at the fourth position of the proline ring introduces specific reactivity patterns that are leveraged during complex organic synthesis. The hydrochloride salt form enhances stability and solubility, facilitating smoother reaction kinetics in multi-step synthesis pathways. It appears as a white powder, indicating high levels of refinement and minimal impurity content. The structural integrity is maintained through controlled crystallization processes.
Industrial Applications
The primary application of this intermediate lies in the production of Zofenopril calcium, a well-known angiotensin-converting enzyme (ACE) inhibitor used globally for managing hypertension and heart failure. The stereochemistry of the (4S) configuration is non-negotiable for the final drug's activity, making the optical purity of this intermediate a critical quality attribute. Pharmaceutical manufacturers rely on this building block to maintain the therapeutic profile of their final formulations. It serves as a key precursor in establishing the sulfur-containing backbone required for the drug's mechanism of action. Beyond Zofenopril, this structural motif is investigated for potential use in other peptidomimetic compounds where proline scaffolds are required to induce specific conformational constraints. The versatility of the phenylthio group allows for further functionalization, making it a valuable asset in medicinal chemistry research and development pipelines focused on cardiovascular health.
Quality Assurance and Specifications
We prioritize industrial purity with an assay specification of ≥98.0%. Each batch undergoes rigorous analytical testing, including High-Performance Liquid Chromatography (HPLC) and Nuclear Magnetic Resonance (NMR) spectroscopy, to verify chemical identity and chiral purity. Documentation such as Certificates of Analysis (COA) is provided with every shipment to ensure regulatory compliance and traceability. Our facility operates under guidelines that support Good Manufacturing Practice (GMP) standards, ensuring that the material is suitable for use in regulated pharmaceutical environments. Quality assurance teams monitor every stage of production, from raw material sourcing to final packaging, to prevent contamination. This commitment to excellence minimizes batch-to-batch variation, providing our clients with the consistency needed for validated manufacturing processes.
| Parameter | Value |
|---|---|
| Molecular Formula | C11H14ClNO2S |
| Molecular Weight | 259.752 |
| Appearance | White powder |
| Assay | ≥98.0% |
| CAS Number | 105107-84-4 |
Storage and Logistics
Packaging is flexible, typically supplied in 25 kg drums, but can be customized according to specific customer requirements to optimize logistics and storage. Proper storage in a cool, ventilated place is recommended to maintain stability over time, protecting the compound from moisture and heat degradation. As a global manufacturer, we offer competitive bulk pricing and reliable supply chain solutions to support large-scale production needs. Partnering with us ensures access to high-quality intermediates backed by technical support and consistent availability. We understand the critical nature of supply chain continuity in the pharmaceutical sector and maintain strategic stock levels to meet urgent demands without compromising on quality standards.



