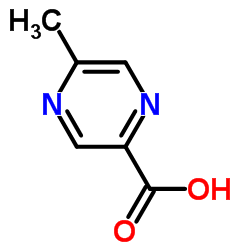
5-Methyl-2-pyrazinecarboxylic acid
- CAS No.5521-55-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 5-Methyl-2-pyrazinecarboxylic acid (CAS 5521-55-1) serves as a critical pharmaceutical intermediate for Glipizide synthesis, available in bulk quantities with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
5-Methyl-2-pyrazinecarboxylic acid is a specialized organic compound widely recognized for its pivotal role in the pharmaceutical industry. As a key building block, this chemical facilitates the synthesis of advanced therapeutic agents, most notably serving as an essential intermediate in the production of Glipizide. Our manufacturing process ensures that every batch meets rigorous international standards, providing clients with a reliable foundation for their drug development pipelines.
We understand the critical nature of supply chain consistency in pharmaceutical manufacturing. Therefore, our facility employs state-of-the-art synthesis routes to maintain high industrial purity levels. This compound is characterized by its stable pyrazine ring structure, which offers excellent reactivity for downstream coupling reactions. Whether for research-scale development or large-scale commercial production, our material provides the consistency required for regulatory compliance.
Key Specifications
Quality assurance is the cornerstone of our operation. Each lot undergoes comprehensive analysis to verify physical and chemical properties. The following table outlines the standard specifications and typical test results associated with our premium grade material.
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | Almost white or light yellow powder | Conform |
| Melting point | 163-167℃ | 166.0℃ |
| Moisture | ≤0.5% | 0.13% |
| Assay | ≥99.0% | 99.84% |
| Single impurity | ≤0.5% | Conform |
| Total impurities | ≤1.0% | 0.16% |
| Conclusion | Conforms to Factory Standard | |
Industrial Applications
The primary application of 5-Methyl-2-pyrazinecarboxylic acid lies in the synthesis of antidiabetic medications. Its high purity profile ensures minimal side reactions during the manufacturing process, thereby optimizing yield and reducing waste. Beyond its use in Glipizide production, this chemical serves as a versatile intermediate for various heterocyclic compound syntheses. Researchers and process chemists value this material for its predictable performance in complex organic transformations.
Our technical team supports clients with detailed manufacturing process documentation. We recognize that different synthesis routes may require specific particle sizes or moisture levels. Consequently, we offer customizable processing options to align with your specific formulation requirements. This flexibility allows for seamless integration into existing production lines without the need for extensive requalification.
Quality Assurance and Logistics
Every shipment is accompanied by a comprehensive Certificate of Analysis (COA), validating the identity and purity of the product. Our quality control laboratory utilizes advanced chromatography and spectroscopy methods to detect impurities at trace levels. This commitment to transparency builds trust with regulatory bodies and ensures patient safety in the final pharmaceutical product.
We operate as a global manufacturer with robust logistics capabilities. Products are packaged in 25kg cardboard drums or according to customer-specified requirements to ensure integrity during transit. Our warehouse facilities maintain strict environmental controls to prevent degradation prior to shipment. For bulk price inquiries or technical support regarding synthesis routes, our sales team is ready to assist with prompt quotations and sample availability.
Storage and Handling
To maintain optimal stability, 5-Methyl-2-pyrazinecarboxylic acid should be stored in a cool and dry well-closed container. It is imperative to keep the material away from moisture and strong light or heat sources, as these factors can influence physical properties over extended periods. Proper handling procedures include using appropriate personal protective equipment during weighing and transfer operations. Adhering to these storage guidelines ensures that the chemical retains its specified purity and performance characteristics until use.



