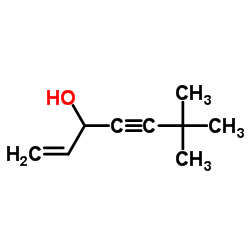
6,6-Dimethylhept-1-en-4-yn-3-ol
- CAS No.78629-20-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate for Terbinafine synthesis. Available in bulk with strict quality control and global logistics support.
Request Bulk PricingProduct Technical Details
Product Overview
6,6-Dimethylhept-1-en-4-yn-3-ol is a critical organic synthesis intermediate characterized by its unique en-yne alcohol structure. This compound serves as a fundamental building block in the pharmaceutical industry, specifically designed for the production of advanced antifungal agents. Our manufacturing facility employs state-of-the-art chemical engineering processes to ensure consistent molecular integrity and high batch-to-batch reproducibility. As a dedicated supplier of fine chemicals, we prioritize the delivery of materials that meet rigorous international standards for pharmaceutical synthesis.
Technical Specifications
Quality control is paramount in the production of pharmaceutical intermediates. Each batch undergoes comprehensive analytical testing to verify compliance with specified parameters. The following table outlines the key physical and chemical properties guaranteed for this product:
| Parameter | Specification |
|---|---|
| CAS Number | 78629-20-6 |
| Molecular Formula | C9H14O |
| Molecular Weight | 138.21 g/mol |
| Appearance | Colorless Liquid |
| Purity | ≥99.0% |
| Density | 0.9 g/cm³ |
| Boiling Point | 187ºC |
| Refractive Index | 1.472 |
| Moisture Content | <1.0% |
| Heavy Metals | <0.001% |
Industrial Applications
The primary application of 6,6-Dimethylhept-1-en-4-yn-3-ol is in the synthesis of Terbinafine and its hydrochloride salt. This intermediate plays a vital role in constructing the specific carbon skeleton required for the final active pharmaceutical ingredient. Our product is optimized for downstream processing, ensuring smooth reaction kinetics and high yields during the coupling and functionalization stages. Pharmaceutical manufacturers rely on our material to maintain the efficiency and safety profiles of their final drug formulations.
Quality Assurance and Safety
We adhere to strict quality management systems to guarantee product safety and reliability. Our laboratory is equipped with advanced chromatography and spectroscopy instruments to detect impurities at trace levels. Safety data sheets are provided with every shipment, outlining proper handling procedures. The compound should be handled in a well-ventilated area with appropriate personal protective equipment. We recommend consulting our technical team for specific safety guidelines related to your manufacturing environment.
Packaging and Storage
To preserve chemical stability, the product is packaged in secure 25kg cardboard drums or customized containers based on client requirements. Storage conditions are critical; the material must be kept in a cool, dry, and well-closed container away from moisture and strong light or heat sources. Proper storage ensures the longevity of the product and maintains its specified purity levels until use. We offer global logistics solutions to ensure timely delivery while maintaining the integrity of the supply chain.



