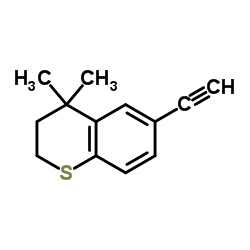
6-Ethynyl-4,4-dimethyl-2,3-dihydrothiochromene
- CAS No.118292-06-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in the synthesis of Tazarotene. Available in bulk with strict quality control and global shipping.
Request Bulk PricingProduct Technical Details
Product Overview
Our organization specializes in the supply of advanced pharmaceutical intermediates, including 6-Ethynyl-4,4-dimethyl-2,3-dihydrothiochromene. This sophisticated organic compound serves as a critical building block in the synthesis of complex therapeutic agents designed for dermatological applications. Engineered for rigorous industrial use, our material undergoes comprehensive quality control protocols to ensure exceptional consistency and reliability for downstream manufacturing processes.
As a key precursor in the pharmaceutical industry, this thiochromene derivative is manufactured to meet the demanding specifications required for active pharmaceutical ingredient synthesis. We understand the vital importance of chemical purity and structural integrity in multi-step synthesis, which is why our production facilities adhere to international safety and quality management standards. Our commitment to excellence ensures that clients receive a product that facilitates efficient reaction pathways.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 118292-06-1 |
| Molecular Formula | C13H14S |
| Molecular Weight | 202.32 g/mol |
| Appearance | White Powder |
| Purity | ≥99.0% |
| Melting Point | 69-72°C |
| Boiling Point | 299°C |
| Density | 1.09 g/cm3 |
Industrial Applications
This chemical intermediate is primarily utilized in the synthesis of Tazarotene, a well-known retinoid used in professional dermatological treatments for psoriasis and acne. The ethynyl group present in the molecular structure facilitates specific coupling reactions that are essential for constructing the final API architecture. Our product ensures high reaction yields and minimizes impurity profiles, which is crucial for maintaining the efficacy and safety of the final pharmaceutical product delivered to patients.
- Essential precursor for dermatological API synthesis.
- Optimized for scalable manufacturing processes.
- Compatible with standard organic synthesis protocols.
- Supports the production of high-performance therapeutic agents.
- Reduces downstream purification burdens.
Quality Assurance and Packaging
We are deeply committed to delivering products that meet the highest industry standards for pharmaceutical intermediates. Each production batch is accompanied by a comprehensive Certificate of Analysis detailing test results from advanced instrumentation such as HPLC and NMR. Our packaging solutions are specifically designed to protect the chemical integrity during international transit, typically available in 25kg cardboard drums or customized according to specific client requirements.
Storage and Handling
To maintain optimal stability and performance, this product should be stored in a cool, dry, and well-closed container. It is imperative to keep the material away from moisture, strong light, and excessive heat sources which could degrade the chemical structure. Proper handling procedures should be followed to ensure safety in the laboratory or industrial setting. Our technical team provides full support on safety data sheets and handling guidelines upon request to ensure compliance with local regulations.



