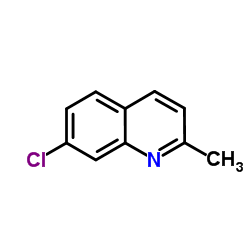
7-Chloro-2-methylquinoline
- CAS No.4965-33-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 7-Chloro-2-methylquinoline (CAS 4965-33-7) designed for pharmaceutical synthesis. Reliable supply for Montelukast intermediate production with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
7-Chloro-2-methylquinoline represents a critical heterocyclic building block within the modern pharmaceutical landscape. As a specialized quinoline derivative, this compound serves as an essential precursor in the multi-step synthesis of advanced therapeutic agents. Our facility produces this material under strict Good Manufacturing Practice guidelines, ensuring consistency and reliability for downstream pharmaceutical manufacturing processes. The chemical structure features a chloro substitution at the seventh position and a methyl group at the second position of the quinoline ring, providing specific reactivity profiles required for complex organic transformations. Furthermore, the molecular stability of this compound allows for extended shelf life when stored correctly, reducing waste and optimizing inventory management for our partners.
Technical Specifications
| Property | Specification |
|---|---|
| CAS Number | 4965-33-7 |
| Molecular Formula | C10H8ClN |
| Molecular Weight | 177.63 g/mol |
| Appearance | White or off-white powder |
| Purity | ≥99% |
| Melting Point | 74-78 °C |
| Boiling Point | 87 °C / 0.5mmHg |
| Density | 1.225 g/cm3 |
Quality Assurance and Control
Quality is the cornerstone of our manufacturing philosophy. Every batch of 7-Chloro-2-methylquinoline undergoes rigorous analytical testing using high-performance liquid chromatography and gas chromatography methods. We maintain a purity level exceeding ninety-nine percent, with impurity profiles meticulously monitored to meet international pharmacopoeia standards. Our quality control team issues a Certificate of Analysis with every shipment, providing full transparency regarding physical properties and chemical composition. This commitment to quality ensures that our clients encounter no disruptions in their own synthesis workflows. We adhere to strict environmental and safety protocols during production, minimizing ecological impact while maximizing yield.
Industrial Applications
This intermediate is predominantly utilized in the production of leukotriene receptor antagonists, specifically serving as a key starting material for Montelukast synthesis. Beyond respiratory therapeutics, quinoline derivatives find applications in various medicinal chemistry programs targeting inflammatory pathways. Our technical support team can provide guidance on optimal storage conditions and handling procedures to maintain stability during your manufacturing process. We understand the critical nature of supply chain continuity in the pharmaceutical industry and offer scalable production capacities to meet both pilot-scale and commercial-scale demands. For clients requiring specific particle size distributions or custom specifications, our technical department offers tailored solutions to align with your specific process requirements.
Packaging and Storage
To preserve integrity, the product is packaged in durable cardboard drums containing twenty-five kilograms net weight, though custom packaging solutions are available upon request. Storage recommendations include keeping the container tightly closed in a cool, dry environment away from direct sunlight and moisture. Proper handling procedures should be followed to ensure safety during transport and warehousing. We coordinate global logistics to ensure timely delivery while maintaining compliance with international chemical shipping regulations. Partnering with us means gaining access to a reliable source of high-performance chemical intermediates backed by decades of industry expertise. Regulatory documentation support is available to facilitate smooth import and export procedures across different jurisdictions.



