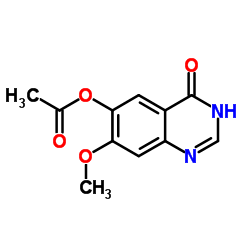
(7-Methoxy-4-oxo-1H-quinazolin-6-yl) Acetate
- CAS No.179688-53-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate for Gefitinib synthesis. Available in bulk with full COA support.
Request Bulk PricingProduct Technical Details
Product Overview
Welcome to our premium selection of advanced pharmaceutical intermediates. The (7-Methoxy-4-oxo-1H-quinazolin-6-yl) Acetate represents a critical building block in the synthesis of modern oncology therapeutics. As a specialized quinazoline derivative, this compound is engineered to meet the rigorous demands of pharmaceutical manufacturing processes. Our facility ensures that every batch undergoes strict quality control protocols to guarantee consistency and reliability for downstream applications.
This chemical entity serves as a vital precursor in the production of tyrosine kinase inhibitors. The structural integrity of the quinazoline scaffold is maintained through optimized synthesis routes, ensuring high reactivity and minimal impurity profiles. We understand the importance of supply chain stability in the pharmaceutical industry, which is why we maintain robust inventory levels to support large-scale production requirements globally.
Technical Specifications
Our commitment to quality is reflected in the detailed physical and chemical properties of this product. We utilize high-performance liquid chromatography (HPLC) to verify purity levels, ensuring that the material meets or exceeds industry standards for pharmaceutical intermediates. The following table outlines the key specification parameters verified during our quality assurance process.
| Parameter | Specification |
|---|---|
| CAS Number | 179688-53-0 |
| Molecular Formula | C11H10N2O4 |
| Molecular Weight | 234.21 g/mol |
| Purity (HPLC) | ≥98.0% |
| Appearance | Off-white to grey to brownish grey powder |
| Density | 1.39 g/cm3 |
| Melting Point | 26°C |
| Boiling Point | 452.39°C |
| Flash Point | 227.40°C |
Industrial Applications
The primary application of this intermediate lies in the synthesis of Gefitinib, a well-known epidermal growth factor receptor (EGFR) inhibitor used in cancer treatment. The precise incorporation of the methoxy and acetoxy functional groups is essential for the biological activity of the final active pharmaceutical ingredient (API). Researchers and process chemists rely on this intermediate for its consistent performance in coupling reactions and subsequent cyclization steps.
Beyond its specific role in Gefitinib production, this quinazoline derivative is also valuable for research and development in medicinal chemistry. It serves as a reference standard for analytical method validation and is used in the exploration of novel kinase inhibitors. Our team works closely with clients to understand their specific synthetic pathways, offering technical support to optimize yield and efficiency.
Quality Assurance and Compliance
We operate under a strict quality management system that aligns with international pharmaceutical standards. Each production batch is accompanied by a comprehensive Certificate of Analysis (COA), detailing test results for identity, purity, and physical properties. We employ advanced analytical instrumentation, including NMR and Mass Spectrometry, to confirm molecular structure and detect any trace impurities.
Compliance with intellectual property rights is a core principle of our business. We adhere to all relevant patent regulations and do not offer products to regions where valid patents may restrict commercial use. Our legal and compliance teams continuously monitor global regulatory landscapes to ensure that all transactions remain within legal boundaries.
Storage and Handling
To maintain the stability and integrity of the chemical, proper storage conditions are essential. The product should be stored in a cool, ventilated place away from direct sunlight and moisture. Containers must be kept tightly sealed when not in use to prevent degradation or contamination. We recommend following standard laboratory safety protocols when handling this material, including the use of appropriate personal protective equipment.
Packaging is available in 25 kg drums, with customization options available upon request to suit specific logistical needs. Our logistics partners ensure safe and timely delivery to destinations worldwide, maintaining the required environmental conditions during transit. For bulk orders or custom synthesis requirements, please contact our sales team for a detailed quotation and lead time information.



