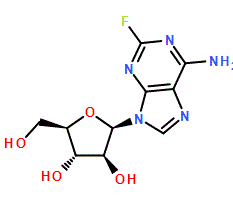
Fludarabine
- CAS No.21679-14-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Fludarabine (CAS 21679-14-1) for pharmaceutical manufacturing. Verified ≥99.0% assay with full documentation and global shipping available.
Request Bulk PricingProduct Technical Details
Product Overview
Fludarabine, also known commercially as F-Ara-A, represents a critical nucleoside analog utilized extensively within the pharmaceutical industry for the development of antineoplastic therapies. Our facility specializes in the production and supply of this high-value chemical entity, ensuring that every batch meets stringent international quality standards. As a leading B2B supplier, we understand the vital importance of consistency and purity in active pharmaceutical ingredients. This product is designed specifically for licensed pharmaceutical manufacturers and research institutions requiring reliable raw materials for formulation and development processes.
The chemical structure, defined as (2R,3S,4S,5R)-2-(6-amino-2-fluoropurin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol, demands precise synthesis protocols. We employ advanced manufacturing processes to maintain the stereochemical integrity of the molecule. Our commitment to quality ensures that the material provided is suitable for downstream processing into final medicinal products. Clients rely on our supply chain stability to maintain their own production schedules without interruption.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 21679-14-1 |
| Molecular Formula | C10H12FN5O4 |
| Molecular Weight | 285.23 g/mol |
| Appearance | White or off-white crystalline powder |
| Assay (HPLC) | ≥99.0% |
| Loss on Drying | ≤1.0% |
| Melting Point | 265-268°C |
Quality Assurance and Control
Quality is the cornerstone of our operation. Each production lot of Fludarabine undergoes rigorous testing protocols using high-performance liquid chromatography (HPLC) and other analytical methods. We provide a comprehensive Certificate of Analysis (COA) with every shipment, detailing all critical quality attributes. Our manufacturing facilities adhere to strict safety and environmental guidelines, ensuring that the product is handled with the utmost care from synthesis to packaging.
We maintain a robust quality management system that tracks every step of the production process. This traceability allows us to guarantee the identity and purity of the chemical. For our partners, this means reduced risk during their own quality control phases and faster time-to-market for their final formulations. We are committed to continuous improvement and regularly audit our processes to align with evolving global regulatory expectations.
Industrial Applications and Handling
This chemical is primarily applied in the synthesis of antineoplastic formulations. It serves as a key active pharmaceutical ingredient for licensed drug manufacturers. Due to its potent nature, proper handling procedures must be followed by trained personnel equipped with appropriate safety gear. We recommend storage in a cool, ventilated place to maintain stability over time. The material is packaged in 25 kg drums, though custom packaging solutions are available to meet specific logistical requirements.
- High purity suitable for pharmaceutical grade applications
- Stable physical properties for long-term storage
- Comprehensive documentation support for regulatory filings
- Global logistics network for timely delivery
- Technical support available for formulation queries
Our team is dedicated to supporting your manufacturing needs with reliable supply and transparent communication. Contact us today to discuss your requirements for Fludarabine and discover how our premium chemical solutions can enhance your production efficiency.



