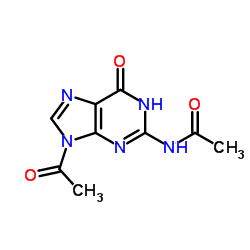
N-(9-Acetyl-6-oxo-3H-purin-2-yl)acetamide
- CAS No.3056-33-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity N-(9-acetyl-6-oxo-3H-purin-2-yl)acetamide (CAS 3056-33-5) for antiviral synthesis. Reliable supply for Acyclovir production with ≥99% assay.
Request Bulk PricingProduct Technical Details
Product Overview
N-(9-acetyl-6-oxo-3H-purin-2-yl)acetamide, frequently recognized by its synonym N2,9-Diacetylguanine, represents a critical high-purity pharmaceutical intermediate within the antiviral medication supply chain. This specialized purine derivative serves as an essential building block in the organic synthesis of Acyclovir, a widely prescribed antiviral agent used to manage herpes simplex virus infections. Our manufacturing facility adheres to strict Good Manufacturing Practices to ensure consistent molecular integrity and batch-to-batch reproducibility. The compound is characterized by its stable crystalline structure and high reactivity in subsequent acylation steps, making it the preferred choice for large-scale pharmaceutical production lines requiring reliable raw material sources.
Technical Specifications
Quality control is paramount in the production of nucleoside analogues. Each batch undergoes rigorous analytical testing using High-Performance Liquid Chromatography to verify purity levels exceeding industry standards. The physical properties are tightly monitored to ensure optimal handling characteristics during downstream processing. Below are the key technical parameters guaranteed for this product:
| Parameter | Specification |
|---|---|
| CAS Number | 3056-33-5 |
| Molecular Formula | C9H9N5O3 |
| Molecular Weight | 235.20 g/mol |
| Appearance | Off-white to light brown powder |
| Assay (HPLC) | ≥99.0% |
| Melting Point | 285°C |
| Density | 1.67 g/cm3 |
Industrial Applications
The primary application of this chemical lies in the pharmaceutical sector, specifically within the synthesis pathway for Acyclovir and related antiviral therapeutics. As a diacetyl protected guanine derivative, it facilitates selective reactions that are crucial for constructing the nucleoside backbone. Pharmaceutical manufacturers rely on this intermediate for its consistent performance in coupling reactions and its minimal impurity profile, which simplifies downstream purification processes. Beyond antiviral drugs, this purine scaffold may also find utility in research and development settings focused on novel nucleotide analogues or biochemical probes. Our supply chain is optimized to support both pilot-scale research projects and commercial-scale manufacturing requirements, ensuring continuity of supply for critical medication production.
Quality Assurance and Packaging
We understand that the integrity of chemical intermediates directly impacts the safety and efficacy of the final drug product. Therefore, every shipment is accompanied by a comprehensive Certificate of Analysis detailing all test results against specifications. The product is packaged in durable 25 kg drums designed to protect against moisture and physical damage during transit. Custom packaging solutions are available upon request to meet specific logistical needs. Storage recommendations include keeping the material in a cool, ventilated environment away from direct sunlight and incompatible substances. Our logistics team ensures global delivery compliance, handling all necessary documentation for international chemical transport. We maintain a robust inventory management system to prevent stockouts and ensure timely fulfillment of urgent orders. Quality assurance protocols extend beyond final product testing to include raw material verification and in-process controls. This multi-layered approach minimizes the risk of contamination and ensures that every gram meets the stringent requirements of pharmaceutical-grade synthesis. For technical inquiries or bulk pricing requests, our sales engineers are available to provide detailed support and sample evaluation. We are committed to fostering long-term partnerships with our clients through transparency, reliability, and scientific expertise.



