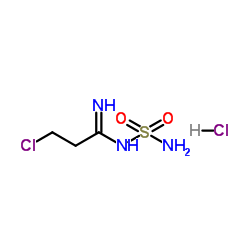
N-Sulphamyl-3-chloropropionamidine Hydrochloride
- CAS No.106649-95-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate specifically designed for the synthesis of Famotidine. Available in bulk quantities with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
N-Sulphamyl-3-chloropropionamidine Hydrochloride represents a critical building block in the modern pharmaceutical landscape, specifically engineered for the production of high-value therapeutic agents. As a key precursor in the synthesis pathway of Famotidine, this compound plays an indispensable role in the manufacturing of H2 receptor antagonists used globally for gastrointestinal treatments. Our facility specializes in the precise organic synthesis of this intermediate, ensuring that every batch meets rigorous international standards for purity and chemical stability.
The molecular structure of this hydrochloride salt is optimized for reactivity in downstream coupling reactions. We understand that the quality of the final active pharmaceutical ingredient depends heavily on the integrity of the starting materials. Therefore, our production process emphasizes strict control over impurities, residual solvents, and physical characteristics to guarantee seamless integration into your manufacturing workflow.
Key Specifications
To ensure transparency and reliability for our B2B partners, we provide detailed analytical data for every production lot. The following table outlines the standard physical and chemical properties expected from our premium grade material.
| Parameter | Specification |
|---|---|
| Chemical Name | N-Sulphamyl-3-chloropropionamidine Hydrochloride |
| CAS Number | 106649-95-0 |
| Molecular Formula | C3H9Cl2N3O2S |
| Molecular Weight | 222.094 g/mol |
| Appearance | White or off-white crystalline powder |
| Assay (HPLC) | ≥98.0% |
| Melting Point | 140-145°C |
| Loss on Drying | ≤1.0% |
| Residue on Ignition | ≤1.0% |
Industrial Applications
This chemical intermediate is primarily utilized in the pharmaceutical industry for the multi-step synthesis of Famotidine. Its specific chloropropionamidine structure allows for efficient nucleophilic substitution and amidine formation reactions required in the final drug assembly. Beyond standard synthesis, this material is suitable for research and development purposes where high-purity reference standards are needed for method validation.
- Primary precursor for Famotidine API manufacturing
- Suitable for large-scale industrial pharmaceutical production
- Compatible with standard GMP synthesis protocols
- Used in quality control laboratories for reference comparison
Quality Assurance and Packaging
Quality is the cornerstone of our manufacturing philosophy. Each batch undergoes comprehensive testing using high-performance liquid chromatography and other validated analytical methods. We provide a Certificate of Analysis with every shipment, detailing the specific test results against our factory standards. Our packaging options are designed to maintain product integrity during transit and storage.
Standard packaging consists of 25 kg fiber drums with inner polyethylene liners to protect against moisture and contamination. Custom packaging solutions are available upon request to suit specific logistical requirements. For optimal stability, we recommend storing the material in a cool, ventilated area away from direct sunlight and strong oxidizing agents. Our supply chain is optimized for global delivery, ensuring timely receipt of materials to keep your production schedules on track.



