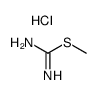
S-Methyl-Isothiourea Hydrochloride
- CAS No.53114-57-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity S-Methyl-Isothiourea Hydrochloride (CAS 53114-57-1) designed for pharmaceutical intermediate synthesis, specifically for Rosuvastatin Calcium production.
Request Bulk PricingProduct Technical Details
Product Overview
S-Methyl-Isothiourea Hydrochloride is a highly specialized organic compound widely recognized for its critical role in modern pharmaceutical synthesis. As a key building block, this chemical serves as an essential intermediate in the production of various therapeutic agents, including statins such as Rosuvastatin Calcium. Our manufacturing process ensures that every batch meets rigorous international standards for purity and consistency, making it a reliable choice for large-scale industrial applications.
We understand the demanding requirements of the pharmaceutical industry. Therefore, our facility employs advanced synthesis techniques to maintain the structural integrity and reactivity of this compound. Whether used in research and development or full-scale commercial production, this product provides the foundational chemistry necessary for efficient downstream processing.
Technical Specifications
| Parameter | Value |
|---|---|
| Chemical Name | S-Methyl-Isothiourea Hydrochloride |
| CAS Number | 53114-57-1 |
| Molecular Formula | C2H7ClN2S |
| Molecular Weight | 126.61 g/mol |
| Purity | ≥98.0% |
| Appearance | White Powder |
| Density | 1.26 g/cm3 |
| Boiling Point | 138.8°C at 760 mmHg |
| Flash Point | 37.7°C |
Industrial Applications
The primary application of S-Methyl-Isothiourea Hydrochloride lies within the pharmaceutical sector, where it acts as a vital precursor in multi-step synthesis routes. Its chemical structure allows for versatile reactivity, facilitating the formation of complex heterocyclic systems found in many active pharmaceutical ingredients (APIs). Specifically, it is utilized in the manufacturing process of cardiovascular medications, demonstrating its importance in producing life-saving therapies.
Beyond specific drug synthesis, this compound is valued for its stability and solubility profiles, which streamline reaction conditions in industrial reactors. Chemical engineers and procurement managers prefer this grade for its predictable performance, reducing the risk of batch failures and ensuring optimal yield rates during production cycles.
Quality Assurance and Storage
Quality control is paramount in our operations. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) verifying all physical and chemical parameters. We adhere to strict Good Manufacturing Practices (GMP) guidelines to ensure product safety and traceability from raw material sourcing to final packaging.
- Packaging: Available in 25kg cardboard drums or customized according to client specifications.
- Storage Conditions: Store in a tightly closed container in a cool, dry, and well-ventilated area.
- Safety: Keep away from incompatible substances and sources of ignition due to flash point considerations.
- Support: Our technical team provides ongoing support for handling guidelines and integration into your specific synthesis routes.
For global partners seeking a dependable supply chain, we offer competitive bulk pricing and logistics solutions tailored to meet regional regulatory requirements. Contact us today to discuss your specific volume needs and request sample data for validation.



