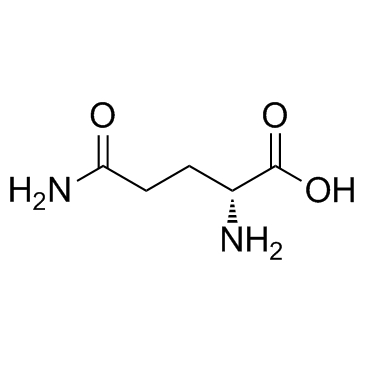
Creatine Monohydrate
- CAS No.6020-87-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Creatine Monohydrate (CAS 6020-87-7) designed for sports nutrition and pharmaceutical formulations. Manufactured under strict GMP standards ensuring superior solubility and stability.
Request Bulk PricingProduct Technical Details
Product Overview
Creatine Monohydrate is a premier nutraceutical ingredient widely recognized for its role in supporting energy metabolism within muscle and brain tissues. As a leading global manufacturer, we provide this essential compound in its most stable and bioavailable form. Our production process adheres to rigorous international quality standards, ensuring that every batch delivers consistent performance for downstream formulation partners. This product serves as a critical component for sports nutrition supplements, clinical nutrition products, and specialized functional foods.
Technical Specifications
Our facility employs advanced crystallization and drying technologies to achieve exceptional purity levels. The following table outlines the key quality parameters verified through our internal quality control laboratories. Each certificate of analysis (COA) is generated per batch to guarantee compliance with specified limits.
| Items of Analysis | Standard | Test Results |
|---|---|---|
| Appearance | White Crystalline Odorless | Conforms |
| Assay | 99.90% Min | 99.93% |
| Loss on Drying | 11.50% Max | 11.21% |
| Creatinine | 50ppm Max | Conforms |
| Heavy Metals | 10ppm Max | Conforms |
| Microbial Count | 10cfu/g Max | Conforms |
| Mesh Size | 80 Mesh | Conforms |
Industrial Applications
This ingredient is primarily utilized in the development of high-performance dietary supplements aimed at enhancing physical output and recovery. Formulators value this material for its excellent stability in dry powder blends and its compatibility with various flavoring systems. Beyond sports nutrition, it is increasingly incorporated into cognitive health formulations due to its role in cellular energy turnover. Our technical team provides comprehensive formulation guides to assist partners in optimizing dosage forms including capsules, tablets, and ready-to-mix powders.
Quality Assurance and Storage
We maintain a robust quality management system certified under GMP guidelines. Raw materials are sourced from approved vendors, and finished products undergo extensive testing for identity, potency, and safety contaminants such as heavy metals and microbiological agents. To maintain optimal quality, we recommend storing the material in a cool, dry environment at room temperature. Containers should remain tightly sealed to prevent moisture absorption, which could impact flow properties during manufacturing. We offer global shipping solutions with full documentation support to ensure seamless supply chain integration for our clients.