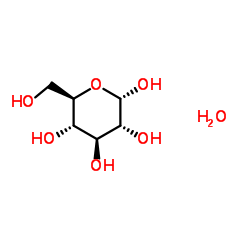
D-Glucose Monohydrate
- CAS No.5996-10-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity D-Glucose Monohydrate USP grade suitable for pharmaceutical injections and nutritional formulations.
Request Bulk PricingProduct Technical Details
Product Overview
D-Glucose Monohydrate is a fundamental carbohydrate compound widely recognized for its critical role in pharmaceutical and nutraceutical industries. As a monohydrate form of D-Glucose, this substance offers exceptional stability and solubility, making it an ideal choice for various medical applications including intravenous solutions and oral supplements. Our manufacturing process ensures that every batch meets stringent international standards, providing reliability for downstream formulation developers.
This compound serves as a primary energy source in metabolic processes and is essential for parenteral nutrition. The high purity profile guarantees safety for human consumption, adhering to rigorous pharmacopoeial requirements. We specialize in supplying bulk quantities to global partners, ensuring consistent quality and timely delivery for large-scale production needs.
Key Specifications
| Parameter | Specification |
|---|---|
| Molecular Formula | C6H14O7 |
| Molecular Weight | 198.17 g/mol |
| CAS Number | 5996-10-1 |
| Appearance | Colorless crystal or white crystalline powder |
| Assay | 97.5% to 102.0% |
| Water Content | 7.5% to 9.5% |
| Related Substances | Total Impurities ≤ 0.5% |
| Conductivity | ≤ 20 μS/cm |
| Microbial Limits | Meets USP Requirements |
Industrial Applications
- Pharmaceutical Injections: Used as a primary component in glucose injections and glucose sodium oxide injections for energy supplementation.
- Nutraceutical Formulations: Serves as a base ingredient for dietary supplements and sports nutrition products.
- Clinical Nutrition: Essential for parenteral feeding solutions in hospital settings.
- Food Industry: Utilized as a sweetener and texturizer in various processed food items.
Quality Assurance
Quality is the cornerstone of our production philosophy. Each batch of D-Glucose Monohydrate undergoes comprehensive testing to ensure compliance with USP43 standards. Our analytical procedures cover identification, assay purity, related substances, and microbial limits. We maintain a robust quality management system that tracks every step from raw material sourcing to final packaging.
Our facility operates under strict GMP guidelines, ensuring that the product remains free from contaminants such as dextrin, soluble starch, and sulfites. Regular audits and third-party validations confirm our commitment to excellence. Customers receive a detailed Certificate of Analysis with every shipment, providing full transparency on test results including aerobic plate counts and bacterial endotoxin levels.
Global Supply and Logistics
We understand the importance of supply chain reliability in the pharmaceutical sector. Our logistics network supports global distribution, ensuring that products arrive in optimal condition. Packaging options are customizable to meet specific client requirements, ranging from standard drums to bulk containers. Partner with us for a secure, compliant, and efficient sourcing experience for your nutraceutical and pharmaceutical needs.