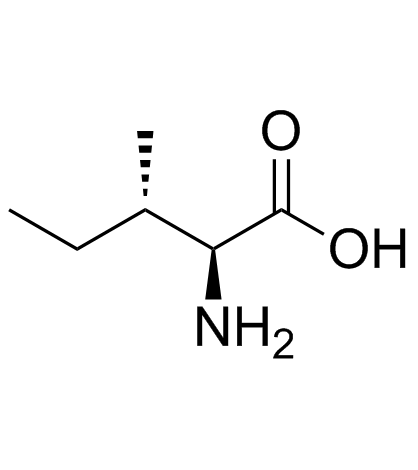
L-Isoleucine
- CAS No.73-32-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Premium L-Isoleucine (CAS 73-32-5) is an essential branched-chain amino acid manufactured for nutraceutical and pharmaceutical applications.
Request Bulk PricingProduct Technical Details
Product Overview
L-Isoleucine is a critical essential amino acid that plays a fundamental role in human nutrition and metabolic processes. As a key member of the branched-chain amino acids (BCAAs) group, it is indispensable for muscle protein synthesis and energy regulation. Our high-purity L-Isoleucine is produced under strict quality control standards to meet the rigorous demands of the nutraceutical and pharmaceutical industries. This compound is widely recognized for its stability and bioavailability, making it an ideal choice for various formulations including dietary supplements, clinical infusions, and specialized infant nutrition products.
We understand the importance of stereochemical purity in amino acid applications. Our manufacturing process ensures the exclusive production of the L-isomer, specifically the (2S,3S) configuration, which is the biologically active form required for human metabolism. This precision guarantees optimal performance in downstream applications where consistency and efficacy are paramount.
Technical Specifications
Our L-Isoleucine complies with major international pharmacopeia standards, ensuring reliability across global markets. The following table outlines the key physical and chemical properties of our standard grade product.
| Parameter | Specification |
|---|---|
| CAS Number | 73-32-5 |
| Assay (Dry Basis) | 99.0% - 101.0% |
| Appearance | White crystals or crystalline powder |
| Odor and Taste | Odorless or slightly characteristic, slightly bitter |
| Specific Rotation [α]20D | +39.5° to +41.5° |
| pH Value (1% Solution) | 5.5 - 6.5 |
| Loss on Drying | Not More Than 0.20% |
| Residue on Ignition | Not More Than 0.10% |
Quality and Purity Control
Quality assurance is the cornerstone of our production facility. Each batch of L-Isoleucine undergoes comprehensive testing to verify purity and safety. Impurity profiles are strictly monitored to ensure compliance with safety limits for heavy metals, residual solvents, and microbial contaminants.
| Impurity Test | Limit |
|---|---|
| Ammonium (NH4) | Not More Than 0.020% |
| Chloride (Cl) | Not More Than 0.020% |
| Sulfate (SO4) | Not More Than 0.020% |
| Iron (Fe) | Not More Than 10 ppm |
| Heavy Metals (Pb) | Not More Than 10 ppm |
| Arsenic (As2O3) | Not More Than 1 ppm |
| Related Substances | Not More Than 0.5% |
| Endotoxin | Less Than 6.0 EU/g |
Our facility is equipped to provide customized grades upon request, including FCC grade with reduced lead content and endotoxin-certified batches for sensitive parenteral applications. All products meet the residual solvent requirements listed in the current JP, USP, and EP monographs.
Industrial Applications
Due to its essential nature and high safety profile, L-Isoleucine is utilized across several high-value sectors. In the pharmaceutical industry, it is a vital component in infusion solutions designed for patient nutrition support. In the nutraceutical sector, it is a primary ingredient in sports nutrition formulations and dietary supplements aimed at muscle recovery and growth. Additionally, its high purity makes it suitable for use in cell culture media, where consistent amino acid profiles are critical for research and bioproduction outcomes.
Packaging and Storage
To maintain product integrity, L-Isoleucine is packaged in secure, moisture-resistant containers. Standard packaging options include 25kg and 50kg drums or bags, tailored to meet client logistics requirements. Storage recommendations include keeping the product in a tight container at controlled room temperature, away from direct sunlight and excessive humidity. Proper storage ensures the long-term stability of the amino acid profile and physical characteristics.