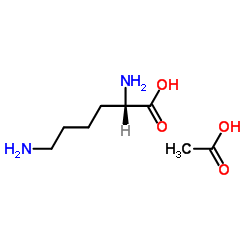
L-Lysine Acetate
- CAS No.57282-49-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity L-Lysine Acetate suitable for nutraceutical formulations and medical infusions, compliant with USP/EP/JP standards.
Request Bulk PricingProduct Technical Details
Product Overview
L-Lysine Acetate represents a premium grade acetate salt of L-lysine, an essential amino acid critical for human metabolic processes. As a fundamental building block for protein synthesis, this compound plays a vital role in various physiological functions, including tissue repair and enzyme production. Our manufactured grade is specifically optimized for high solubility and stability, making it an ideal choice for complex pharmaceutical and nutraceutical formulations. The acetate salt form enhances bioavailability compared to standard base forms, ensuring efficient absorption in biological systems.
Technical Specifications
We adhere to strict quality control protocols to ensure every batch meets international pharmacopeia standards. The following table outlines the key physical and chemical parameters verified during our quality assurance process.
| Parameter | Specification | Typical Result |
|---|---|---|
| Appearance | White crystals or crystalline powder | Conforms |
| Identification | Infrared Absorption | Conforms |
| Assay | 98.0% to 102.0% | 99.9% |
| Specific Rotation | +8.4 degrees to +9.9 degrees | +8.9 degrees |
| Loss on Drying | Less than or equal to 0.2% | 0.08% |
| Residue on Ignition | Less than or equal to 0.4% | 0.04% |
| Heavy Metals | Less than or equal to 15ppm | Less than 15ppm |
| Microbial Limits | Total viable count less than 1000cfu/g | Conforms |
Industrial Applications
This high-purity amino acid salt is extensively utilized in the preparation of parenteral nutrition infusions, where sterility and precise composition are paramount. Beyond clinical settings, it serves as a key fortification ingredient in dietary supplements designed to support muscle recovery and immune function. The material is also compatible with various liquid and solid dosage forms, providing formulators with flexibility in product development. Our endotoxin-certified grades are available upon specific request for sensitive applications requiring ultra-low pyrogen levels.
Quality Assurance and Storage
Production is conducted in facilities compliant with current Good Manufacturing Practices (cGMP). Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) verifying compliance with USP, EP, and JP monographs regarding residual solvents and impurities. To maintain integrity, the product should be stored in tight containers at controlled room temperature, protected from moisture and direct light. We offer flexible packaging options, including 25kg and 50kg drums, to accommodate diverse supply chain requirements for global manufacturers.