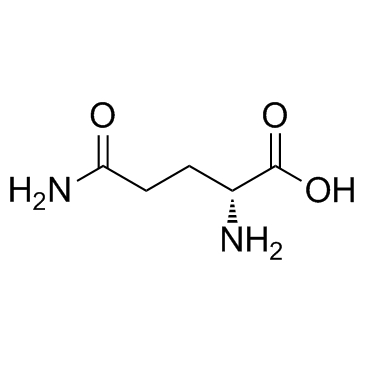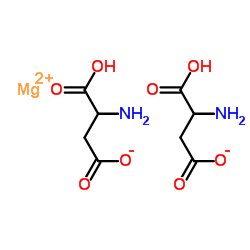
L-Magnesium Aspartate
- CAS No.2068-80-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Premium L-Magnesium Aspartate is a highly bioavailable mineral source designed for nutraceutical formulations. It offers superior solubility and stability for dietary supplements and functional foods.
Request Bulk PricingProduct Technical Details
Product Overview
L-Magnesium Aspartate represents a superior form of mineral supplementation, combining the essential macro-mineral magnesium with the amino acid L-aspartic acid. This organic compound is engineered to maximize bioavailability, ensuring that the body can efficiently absorb and utilize magnesium for critical physiological functions. As a key nutraceutical ingredient, it serves as a foundational component for health products aimed at supporting energy metabolism, muscle function, and nervous system health.
Our manufacturing process adheres to strict international quality standards, producing a high-purity substance that meets the requirements for pharmaceutical and food-grade applications. The chelation of magnesium with aspartate enhances solubility in aqueous solutions, making it an ideal choice for liquid formulations, effervescent tablets, and powdered dietary mixes.
Key Features and Benefits
- Enhanced Bioavailability: The organic salt form facilitates better absorption compared to inorganic magnesium sources.
- High Solubility: Easily soluble in water, allowing for versatile application in various formulation types.
- Physiological Synergy: Aspartic acid plays a role in the Krebs cycle, potentially supporting energy production alongside magnesium.
- Quality Assurance: Available in multiple grades including BP2009, Export Pharmaceutical, and Feed Grade to suit diverse industry needs.
- Stability: Excellent physical stability ensures a long shelf life under proper storage conditions.
Technical Specifications
We maintain rigorous quality control protocols to ensure consistency across all batches. The following table outlines the key parameters for our standard pharmaceutical and export grades.
| Parameter | Pharmaceutical Grade | Export Grade |
|---|---|---|
| Assay (as anhydrous) | 98.0% - 102.0% | 98.0% - 102.0% |
| Specific Rotation | -6.5° | -6.5° |
| pH Value (2.5% solution) | 6.0 - 8.0 | 6.0 - 8.0 |
| Chlorides (Cl) | ≤ 0.05% | ≤ 0.05% |
| Sulphates (SO4) | ≤ 0.1% | ≤ 0.1% |
| Heavy Metals (Pb) | ≤ 0.001% | ≤ 0.001% |
| Water Content | 10.0% - 14.0% | 10.0% - 14.0% |
Industrial Applications
This ingredient is primarily utilized in the production of dietary supplements and functional foods. It is particularly valued in formulations designed to address magnesium deficiency, support athletic performance, or promote cardiovascular health. Due to its favorable taste profile and solubility, it is frequently incorporated into nutraceutical powders, capsules, and fortified beverages. Manufacturers seeking a reliable drop-in replacement for standard magnesium sources will find this material offers performance benchmarks that align with high-end consumer expectations.
Storage and Handling
To maintain product integrity, L-Magnesium Aspartate should be stored in a dry, cool, and well-ventilated warehouse. Protection against dampness is critical to prevent caking or hydrolysis. The standard packaging consists of 25kg kraft paper bags lined with double-layer plastic bags, ensuring protection during global transit. We recommend conducting incoming quality checks upon receipt to verify compliance with the provided Certificate of Analysis (COA).