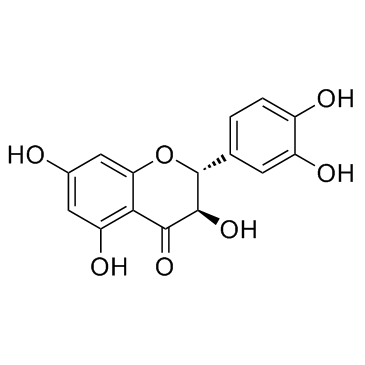
Taxifolin
- CAS No.480-18-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Taxifolin powder (CAS 480-18-2) suitable for nutraceutical and research applications. Manufactured with strict quality control and comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
Taxifolin, frequently recognized in the industry as Dihydroquercetin, is a naturally occurring flavonoid characterized by its potent antioxidant properties. This high-value chemical intermediate is extensively utilized across the nutraceutical and cosmetic sectors due to its stability and bioactive potential. Our manufacturing process ensures that every batch meets rigorous international standards for purity and safety, making it an ideal choice for formulators seeking reliable raw materials for health supplements and skincare solutions.
As a specialized chemical supplier, we understand the critical importance of consistency in molecular structure and potency. The compound is chemically defined as (2R,3R)-3,3',4',5,7-Pentahydroxyflavanone, featuring a specific stereochemistry that contributes to its functional efficacy. We provide comprehensive documentation, including Certificates of Analysis (COA), to guarantee transparency and compliance with global regulatory frameworks for ingredient sourcing.
Key Specifications
| Parameter | Specification | Result |
|---|---|---|
| Appearance | Light yellow powder | Conform |
| Purity (HPLC) | ≥98.0% | 98.5% |
| Loss on Drying | ≤5.0% | 0.89% |
| Total Ash | ≤5.0% | 0.10% |
| Heavy Metals (Pb) | ≤0.5ppm | Conform |
| Heavy Metals (As) | ≤0.3ppm | Conform |
| Microbiological Count | ≤1000CFU/g | Conform |
| E. Coli | Negative | ND |
Industrial Applications
This flavonoid ingredient is primarily valued for its capacity to support oxidative balance in various biological systems. In the nutraceutical industry, it is incorporated into dietary supplements designed to promote overall wellness and cellular health. Furthermore, its compatibility with diverse formulation matrices allows for seamless integration into capsules, tablets, and liquid concentrates. In the cosmetic sector, Taxifolin is appreciated for its ability to protect skin formulations from oxidative stress, thereby enhancing product stability and efficacy.
- High antioxidant activity suitable for wellness formulations.
- Excellent solubility profile for diverse delivery systems.
- Strictly controlled heavy metal and microbiological limits.
- Verified structural integrity via advanced spectroscopic analysis.
- Scalable production capacity for bulk industrial requirements.
Storage and Handling
To maintain optimal chemical stability and potency, this product should be stored in a cool, dry environment away from direct sunlight and moisture. We recommend maintaining storage temperatures at -20°C in sealed containers to prevent degradation over extended periods. Proper handling procedures, including the use of personal protective equipment during weighing and processing, ensure safety during manufacturing operations. Our packaging solutions are designed to protect the integrity of the powder during global transit, ensuring it arrives at your facility in pristine condition.