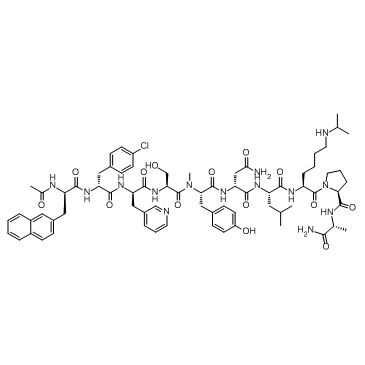
Abarelix
- CAS No.183552-38-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Abarelix API for pharmaceutical formulation. Competitive bulk pricing and reliable supply chain for global manufacturers.
Request Bulk PricingProduct Technical Details
Product Overview
Abarelix represents a sophisticated class of synthetic decapeptide antagonists designed for advanced pharmaceutical applications. As a gonadotropin-releasing hormone (GnRH) antagonist, this compound plays a critical role in the development of specialized therapeutic formulations. Our facility specializes in the production of high-purity peptide active pharmaceutical ingredients (APIs), ensuring that every batch meets rigorous international standards for chemical integrity and stability. This product is intended strictly for licensed pharmaceutical manufacturing and research purposes.
The molecular structure of Abarelix is complex, requiring precise solid-phase peptide synthesis techniques to achieve the desired stereochemistry and biological activity. We employ state-of-the-art purification methods to remove impurities, ensuring a final product that supports consistent downstream formulation performance. Our commitment to quality begins with raw material sourcing and extends through final packaging and logistics.
Technical Specifications
Understanding the physicochemical properties of this peptide is essential for proper handling and integration into manufacturing processes. The following data reflects our standard production specifications, verified through advanced analytical instrumentation.
| Parameter | Value |
|---|---|
| Chemical Name | Abarelix |
| CAS Registry Number | 183552-38-7 |
| Molecular Formula | C72H95ClN14O14 |
| Molecular Weight | 1416.063 g/mol |
| Appearance | White to off-white powder |
| Purity (Assay) | ≥98.0% |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 1688.4±65.0 °C at 760 mmHg |
Quality Assurance and Manufacturing
Quality control is the cornerstone of our peptide manufacturing operation. Each production lot undergoes comprehensive testing using High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry to confirm identity and purity. We maintain strict documentation practices, providing a Certificate of Analysis (COA) with every shipment. Our manufacturing processes are designed to minimize batch-to-batch variability, which is crucial for pharmaceutical clients requiring consistent input materials for regulatory submissions.
- Advanced purification protocols to ensure high assay values.
- Stability testing under various conditions to guarantee shelf life.
- Compliance with international safety and handling guidelines.
- Dedicated support for technical queries and regulatory documentation.
Storage and Handling Guidelines
To maintain the chemical stability of Abarelix, proper storage conditions are mandatory. The material should be kept in a cool, ventilated environment away from direct sunlight and moisture. Containers must remain sealed when not in use to prevent degradation. For large-scale industrial use, we recommend consulting our technical team regarding specific storage infrastructure requirements. We offer flexible packaging solutions, including standard 25 kg drums or customized configurations based on client logistics needs. Our global supply chain ensures timely delivery while maintaining cold chain integrity where necessary.
Commercial Partnership
We understand the critical nature of supply chain reliability in the pharmaceutical industry. Our team is prepared to support long-term contracts with consistent quality and competitive pricing structures. Whether you require small-scale research quantities or large-volume production batches, we have the capacity to meet your demands. Contact our sales department for detailed technical dossiers and customized quotations tailored to your specific project requirements.



