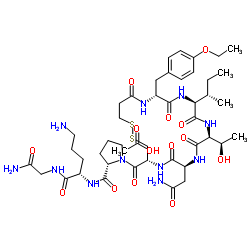
Atosiban Acetate
- CAS No.914453-95-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Atosiban Acetate peptide API for pharmaceutical manufacturing and research applications with verified COA.
Request Bulk PricingProduct Technical Details
Product Overview
Atosiban Acetate represents a high-quality cyclic nonapeptide analogue designed for advanced pharmaceutical development and research purposes. As a competitive antagonist targeting specific neurohypophyseal hormone receptors, this compound is synthesized under stringent quality control protocols to ensure maximum purity and structural integrity. Our manufacturing facility adheres to international standards, providing a reliable supply chain for global partners seeking premium peptide building blocks for complex therapeutic formulations.
The chemical structure of Atosiban Acetate is meticulously engineered to maintain stability during storage and processing. This product serves as a critical active pharmaceutical ingredient (API) or intermediate for laboratories and production units focused on obstetric care solutions. We prioritize consistency in every batch, ensuring that the molecular weight and formula align precisely with theoretical specifications required for downstream synthesis and formulation processes.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 914453-95-5 |
| Molecular Formula | C45H71N11O14S2 |
| Molecular Weight | 1054.240 |
| Appearance | White to off-white powder |
| Purity (HPLC) | ≥98.0% |
| Test Result | 98.15% |
| Conclusion | Conforms to Factory Standard |
Industrial Applications
This peptide acetate salt is primarily utilized in the synthesis of pharmaceutical formulations targeting specific physiological pathways. In the context of B2B chemical supply, it serves as a key component for research institutions and manufacturing entities developing specialized therapeutic agents. The high affinity profile of this molecule makes it suitable for applications requiring precise receptor interaction studies.
Our clients leverage this material for various stages of drug development, from initial preclinical research to large-scale production runs. The versatility of Atosiban Acetate allows it to be integrated into diverse delivery systems, provided that all handling procedures follow established safety guidelines. We support partners who require a dependable source of material that meets rigorous analytical standards for performance benchmarking and equivalent substitution in existing protocols.
Quality Assurance and Storage
Quality is the cornerstone of our operation. Every batch of Atosiban Acetate undergoes comprehensive testing using high-performance liquid chromatography (HPLC) and other advanced analytical methods. We provide a Certificate of Analysis (COA) with every shipment, detailing the specific test results against our factory standards. This transparency ensures that our partners can maintain their own quality management systems without compromise.
To maintain optimal stability, the product should be stored in a cool, ventilated environment away from direct sunlight and moisture. We offer flexible packaging solutions, including standard 25 kg drums, with customization available based on specific logistical requirements. Our global manufacturing network ensures timely delivery while maintaining the cold chain or protective packaging necessary for sensitive peptide materials. For bulk pricing inquiries or technical documentation, our support team is ready to assist with detailed formulation guides and manufacturing process insights.



