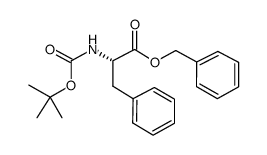
Boc-L-Phe-OBzl
- CAS No.66617-58-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Boc-L-Phe-OBzl (CAS 66617-58-1) is a premium protected amino acid derivative designed for efficient peptide synthesis and pharmaceutical intermediate applications.
Request Bulk PricingProduct Technical Details
Product Overview
Boc-L-Phe-OBzl, chemically known as N-(tert-Butoxycarbonyl)-L-phenylalanine benzyl ester, stands as a critical building block in the realm of organic synthesis and peptide chemistry. Identified by CAS number 66617-58-1, this compound features a phenylalanine backbone protected by a tert-butoxycarbonyl (Boc) group at the N-terminus and a benzyl ester at the C-terminus. This dual protection strategy offers chemists exceptional flexibility during multi-step synthesis protocols, allowing for selective deprotection under mild acidic or hydrogenolytic conditions respectively.
Our manufacturing process ensures that every batch meets rigorous international standards for pharmaceutical intermediates. The compound is synthesized using high-grade raw materials and undergoes extensive purification to eliminate impurities that could interfere with downstream coupling reactions. As a protected amino acid derivative, it serves as a foundational element for constructing complex peptide chains, active pharmaceutical ingredients (APIs), and specialized biochemical research compounds.
Technical Specifications
Quality consistency is paramount in chemical manufacturing. We employ advanced analytical techniques including HPLC, NMR, and Mass Spectrometry to verify the identity and purity of each production lot. The physical characteristics are optimized for ease of handling in both laboratory and industrial scale operations.
| Parameter | Specification |
|---|---|
| CAS Number | 66617-58-1 |
| Molecular Formula | C21H25NO4 |
| Molecular Weight | 355.43 g/mol |
| Appearance | White Crystalline Powder |
| Purity (HPLC) | ≥98.0% |
| Density | 1.129 g/cm3 |
| Boiling Point | 500.2°C at 760 mmHg |
| Flash Point | 256.3°C |
| Refractive Index | 1.547 |
| Packaging | 25 kg/drum or Customized |
Industrial Applications
The primary utility of Boc-L-Phe-OBzl lies in its role as a versatile intermediate for peptide synthesis. The Boc protecting group is stable under basic conditions but can be cleanly removed using trifluoroacetic acid (TFA), making it compatible with various coupling strategies. Meanwhile, the benzyl ester moiety can be removed via catalytic hydrogenation, orthogonal to the Boc group removal. This orthogonality is crucial for solid-phase peptide synthesis (SPPS) and solution-phase fragment condensation.
Beyond peptide construction, this compound is utilized in the development of protease inhibitors, enzyme substrates, and modified amino acid therapeutics. Pharmaceutical companies rely on this intermediate for the production of complex biologics and small molecule drugs where stereochemical integrity is essential. Our product supports both research-scale discovery and large-scale commercial manufacturing needs.
Storage and Handling
To maintain optimal stability and purity, proper storage conditions must be observed. The product should be kept in a tightly sealed container to prevent moisture absorption, which could lead to hydrolysis of the ester bond. Store in a cool, dry, and well-ventilated area away from direct sunlight and strong oxidizing agents. Under recommended storage conditions at room temperature, the shelf life extends significantly while maintaining specified purity levels.
Safety during handling is ensured by following standard laboratory protocols. Personnel should wear appropriate personal protective equipment including gloves and safety goggles. In case of spillage, clean up immediately using appropriate absorbent materials. We provide comprehensive documentation including Certificates of Analysis (COA), Material Safety Data Sheets (MSDS), and method validation reports to support regulatory compliance and quality assurance audits.



