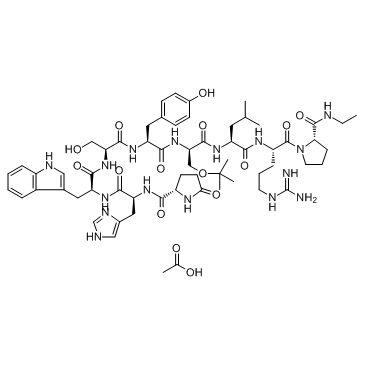
Buserelin Acetate
- CAS No.68630-75-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Buserelin Acetate for pharmaceutical manufacturing. Reliable supply with full documentation and COA.
Request Bulk PricingProduct Technical Details
Product Overview
Buserelin Acetate is a synthetic nonapeptide analog of native gonadotropin-releasing hormone (GnRH). As a potent agonist, it plays a critical role in the pharmaceutical industry for the development of therapeutic formulations. Our facility specializes in the production of high-quality peptide active pharmaceutical ingredients (APIs) designed to meet stringent global regulatory standards. This product is intended for B2B pharmaceutical manufacturing and research applications.
We understand the critical nature of supply chain consistency for peptide-based therapeutics. Our manufacturing process employs advanced solid-phase peptide synthesis techniques followed by rigorous purification steps. This ensures that every batch delivers exceptional consistency, stability, and bioactivity required for downstream drug formulation. We are committed to supporting our partners with reliable volumes and comprehensive technical documentation.
Technical Specifications
Quality control is paramount in the production of complex peptide structures. Each lot undergoes extensive analytical testing to verify identity, purity, and physical characteristics. The following table outlines the key physicochemical properties and quality standards associated with our Buserelin Acetate supply.
| Parameter | Specification |
|---|---|
| CAS Number | 68630-75-1 |
| Molecular Formula | C62H90N16O15 |
| Molecular Weight | 1239.424 g/mol |
| Appearance | White to off-white powder |
| Assay (HPLC) | ≥98.0% |
| Melting Point | 186-188°C |
| Density | 1.4±0.1 g/cm3 |
| Refractive Index | 1.671 |
Industrial Applications
In the pharmaceutical sector, this peptide analog is utilized as a key active ingredient in the formulation of medications targeting hormone-sensitive conditions. It is commonly incorporated into delivery systems such as nasal sprays, subcutaneous implants, or injection solutions. The high purity grade we provide ensures optimal performance in final drug products, minimizing impurities that could affect stability or efficacy.
Our clients leverage this material for developing treatments related to reproductive health and oncology support. By providing a reliable source of high-grade peptide API, we enable pharmaceutical companies to maintain robust production schedules and meet regulatory compliance requirements for market approval. We also support custom packaging solutions to align with specific manufacturing line needs.
Storage and Handling
To maintain the integrity of the peptide structure, proper storage conditions are essential. The material should be kept in a cool, ventilated environment away from direct sunlight and moisture. Containers must remain tightly sealed when not in use to prevent degradation. We recommend storing the product at controlled room temperature or under refrigeration depending on the specific formulation timeline. Always refer to the provided Certificate of Analysis (COA) for batch-specific storage instructions.
Safety data sheets (SDS) are available upon request to ensure safe handling within your facility. Our logistics team ensures secure packaging using industry-standard drums or customized containers to prevent damage during global transit. Contact our sales team for detailed information on lead times, bulk pricing, and regulatory support documentation.



