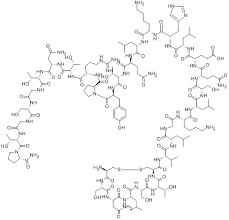
Calcitonin Salmon
- CAS No.47931-85-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Calcitonin Salmon peptide for pharmaceutical manufacturing. GMP compliant supply with full documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Calcitonin Salmon is a highly specialized synthetic polypeptide hormone consisting of 32 amino acids. Structurally analogous to the natural hormone produced in the ultimobranchial body of fish, this compound exhibits significantly higher biological activity compared to human calcitonin. As a critical component in the pharmaceutical industry, it serves as a key active pharmaceutical ingredient (API) for developing therapies related to calcium metabolism and bone health. Our facility produces this peptide under strict quality control standards to ensure consistency and reliability for downstream manufacturing partners.
Designed for professional B2B applications, this product is intended for use in pharmaceutical formulation, clinical research, and advanced biochemical studies. We prioritize regulatory compliance and safety, ensuring that all batches meet rigorous international specifications for purity and identity. Whether utilized for developing injectable solutions or investigative research, our Calcitonin Salmon provides a reliable foundation for high-value therapeutic projects.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 47931-85-1 |
| Molecular Formula | C145H240N44O48S2 |
| Molecular Weight | 3431.853 g/mol |
| Appearance | White to off-white powder |
| Assay (Purity) | ≥98.0% |
| Density | 1.5±0.1 g/cm3 |
| Refractive Index | 1.677 |
| Packing | 25 kg/drum or customized |
Quality Assurance and Manufacturing
Quality is the cornerstone of our production process. Each batch of Calcitonin Salmon undergoes comprehensive analytical testing using high-performance liquid chromatography (HPLC) and mass spectrometry to verify sequence integrity and purity levels. We maintain a robust quality management system that aligns with global Good Manufacturing Practice (GMP) guidelines. This ensures that the peptide remains stable and free from contaminants during storage and transportation.
Our manufacturing capabilities allow for scalable production to meet diverse client demands, from pilot-scale research quantities to large commercial volumes. We provide full documentation support, including Certificates of Analysis (COA), Method Validation Reports, and Stability Data, facilitating seamless regulatory filings for our partners. Traceability is maintained throughout the supply chain, guaranteeing transparency and accountability.
Storage and Handling
To maintain optimal stability and potency, proper storage conditions are essential. The product should be kept in a cool, ventilated environment away from direct sunlight and moisture. Containers must remain tightly sealed when not in use to prevent degradation due to humidity or oxidation. For long-term storage, refrigeration is recommended to preserve the structural integrity of the peptide chain. Handling should be conducted by trained personnel using appropriate personal protective equipment to ensure safety and prevent contamination.
Industrial Applications
- Pharmaceutical formulation for bone metabolism therapies.
- Research and development in endocrinology and calcium regulation.
- Reference standard for analytical laboratories.
- Component in specialized biochemical assays.
By choosing our Calcitonin Salmon, partners gain access to a premium grade peptide backed by decades of chemical synthesis expertise. We are committed to supporting innovation in the life sciences sector through reliable supply chains and technical excellence.



