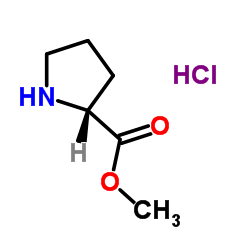
D-Proline Methyl Ester
- CAS No.65365-28-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity chiral building block for pharmaceutical synthesis.
Request a Quote & Full COAProduct Technical Details
Product Overview
D-Proline Methyl Ester represents a critical chiral building block within the realm of advanced organic synthesis and pharmaceutical manufacturing. As a protected amino acid derivative, this compound plays an indispensable role in the construction of complex molecular architectures required for modern therapeutics. Our facility specializes in the production of high-enantiomeric purity intermediates, ensuring that every batch meets the rigorous demands of medicinal chemistry and process development teams worldwide.
This esterified form of D-Proline offers enhanced solubility and reactivity profiles compared to the free acid, making it an ideal candidate for various coupling reactions and cyclization processes. The hydrochloride salt form ensures stability during storage and handling, providing chemists with a reliable reagent for scale-up operations.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | D-Proline Methyl Ester Hydrochloride |
| CAS Number | 65365-28-8 |
| Molecular Formula | C6H12ClNO2 |
| Molecular Weight | 165.62 g/mol |
| Purity | ≥98.0% |
| Appearance | White Crystalline Powder |
| Melting Point | 69-71 °C |
| Loss on Drying | ≤0.5% |
| Heavy Metals | ≤10 ppm |
Industrial Applications
The primary utility of this compound lies in its function as a key precursor for the synthesis of biologically active alkaloids and pharmaceutical active ingredients (APIs). It is extensively utilized in the preparation of chiral anesthetics and complex nitrogen-containing heterocycles. Specific synthetic pathways include the production of local anesthetics such as Ropivacaine, where stereochemical integrity is paramount for safety and efficacy.
- Synthesis of chiral pharmaceutical intermediates
- Construction of pyrrolidine-based alkaloid frameworks
- Development of novel peptidomimetic structures
- Process chemistry for enantiomerically pure drugs
By providing a stable and reactive nitrogen source, this material facilitates the efficient formation of amide bonds and ring-closing reactions essential in multi-step synthesis routes.
Quality Assurance and Control
Quality is the cornerstone of our manufacturing process. Every production lot of D-Proline Methyl Ester undergoes comprehensive analytical testing using high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) spectroscopy. We verify enantiomeric excess to ensure that the chiral integrity remains intact throughout the production cycle. A Certificate of Analysis (COA) is provided with every shipment, detailing all critical quality attributes and confirming compliance with internal specifications.
Storage and Handling
To maintain optimal stability and prevent degradation, this product should be stored in a cool, dry environment at temperatures below -15°C. The container must remain tightly sealed to protect against moisture absorption and atmospheric contamination. When handling, standard laboratory safety protocols should be observed, including the use of appropriate personal protective equipment. We offer flexible packaging solutions, including 25 kg drums, and can accommodate custom packaging requirements to suit specific logistical needs for global distribution.